New Simplified Chemistry Class 10 ICSE Solutions – Organic Chemistry
ICSE SolutionsSelina ICSE SolutionsML Aggarwal Solutions
Viraf J Dalal Chemistry Class 10 Solutions and Answers
Simplified ChemistryEnglishMathsPhysicsChemistryBiology
QUESTIONS
2004
Question 1.
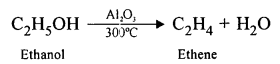
Write the equation for the preparation of ethylene from ethyl alcohol.
Answer:
![]()
Question 2.
State the general formula for a saturated hydrocarbon and give one example and structural formula of the same.
Answer:
General formula for a saturated hydrocarbon (alkanes): CnH2n+2 Example of saturated hydrocarbons (alkanes): C2H2×1+2 Or C1H4
Structural formula of CH4 (Methane)
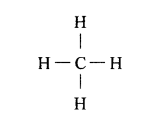
Question 3.
Name a compound which will give acetylene gas when treated with water.
Answer:
CaC2; (calcium carbide)
2005
Question 1.
Define the term ‘catenation’.
Answer:
It is the property of elements by virtue of which atoms of the element can link to each other to form chains or rings of different sizes.
Question 2.
State the term for: – Compounds having the same general formula, and similar chemical properties.
Answer:
Homologous series.
Question 3.
Draw the structural formula of a compound with two carbon atoms in each of the following cases:
- An alkane with a carbon to carbon single
- An alcohol containing two carbon atoms.
- An unsaturated hydrocarbon with a carbon to carbon triple bond
Answer:
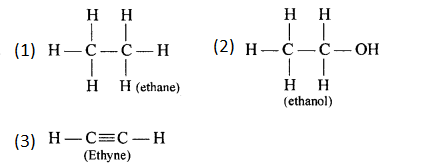
Question 4.
Ethane, Ethene, Ethanoic acid, Ethyne, Ethanol — From the compounds, name :
- The compound with — OH and with — COOH,
- Homologue of homologous series with general formula Cn H2n.
Answer:
- Compound with — OH group: Ethanol (C2H5OH)
- Compound with — COOH group: Ethanoic acid (CH3COOH)
- Homologue of homologous series with G.F. CnH2n:Ethene (C2H4)
Question 5.
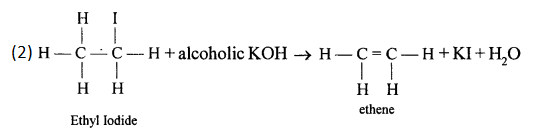
Write the equations for the following laboratory preparations:
- Ethane from Sodium propionate.
- Ethene from Iodoethane.
- Ethyne from Calcium carbide.
- Methanol from Iodomethane.
Answer:
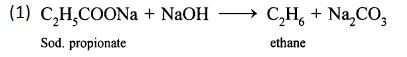
(3) CaC2 + 2H2O →HC≡ CH + Ca(OH)2
2006
Question 1.
Which one of the elements — Li, Be, B, C, O, F, Ne shows the property of catenation.
Answer:
C (carbon).
Question 2.
Write a balanced equation for:
- Reaction of ethane and oxygen in presence of molybdenum oxide.
- Preparation of CH4 from anhydrous sodium ethanoate (sodium acetate).
- Reaction of heating ethanol at 170° C in the presence of cone. H2SO4.
- Preparation of carbon tetrachloride from methane.
Answer:
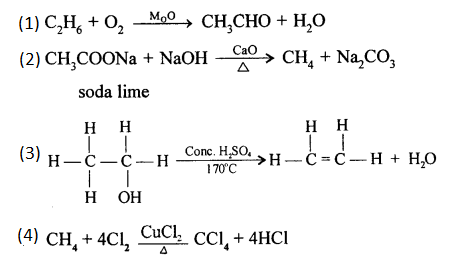
Question 3.
Give the IUPAC name and the functional group for :
- CH3 — CH2 — CHO
- H3C — CH2 — CH2 — OH
Answer:
(1) IUPAC name: Propanal
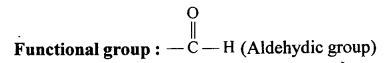
(2) IUPAC name: Propan-l-ol
Functional group: OH (Alcoholic group)
Question 4.
Draw the structural formula of ethyne. How does the structure of alkynes differ from that of alkenes.
Answer:
Structural formula of ethyne
H —C ≡ C —H
In alkynes there are — C ≡ C — triple covalent bonds but in alkenes there are![]() double covalent bonds.
double covalent bonds.
Question 5.
Fill in the blanks with the correct words:
Alkenes are the_______ (analogous / homologous) series of_______ (saturated / unsaturated) hydrocarbons. They differ from alkanes due to the presence of_______ (double / single) bonds. Alkenes mainly undergo________ (addition / substitution) reactions.
Question 6.
Draw the structural formulae of the two isomers of Butane. Give the correct IUPAC name of each.
Answer:
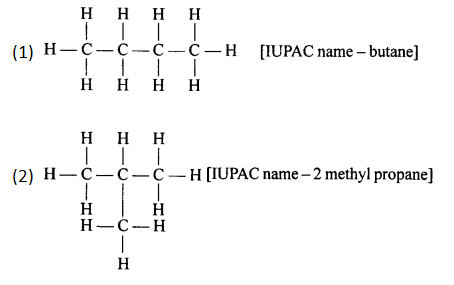
2007
Question 1.
Give the IUPAC names of the compounds numbered (i) to (v).
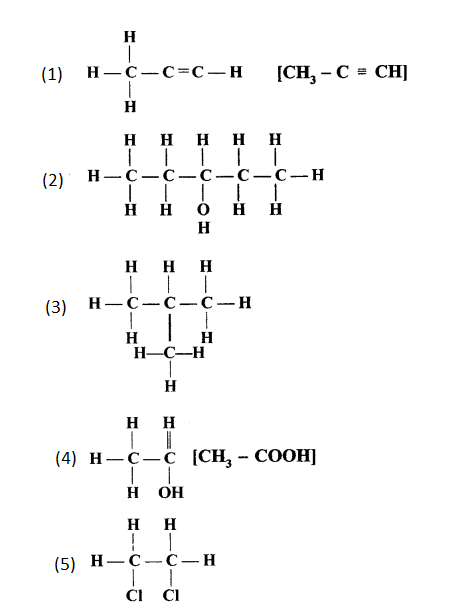
Answer:
- Propyne
- Pentan-3-ol
- 2-Methylpropane
- Ethanoic acid
- 1,2-Dichloroethane
Question 2.
Copland complete the table which relates to three homologous series of Hydrocarbons :

Answer:
- Alkenes, Alkynes and Alkanes
- Double, Triple, Single
- Ethene, Ethyne, Methene
- Addition Substitution
Question 3.
Name the type of reaction by which X (compound of C and Br) can be prepared from ethane.
Answer:
By substitution reactions.
2008
Question 1.
The formation of 1, 2-dibromoethape from ethene and bromine is an example of :
A. Substitution
B. Dehydration
C. Dehydrohalogenation
D. Addition
Question 1.
Name the organic compound prepared by each of the following reactions :
- C2H5COONa + NaOH →
- CH3I + 2[H]-→
- C2H5Br + KOH (alcoholic soln.) →
- CaC2 + 2H2O →
Answer:
(1)
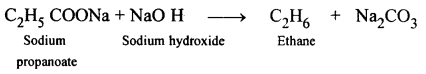
Organic compound formed is Ethane.
(2)
![]()
Organic compound formed is Methane
(3)
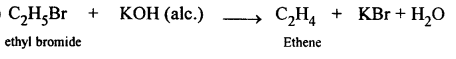
Organic compound formed is Ethene.
(4)
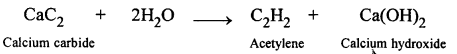
Organic compound formed is Acetylene.
Question 3.
Write the equation for the following :
- Calcium carbide and water
- Ethene and water (steam)
- Bromoethane and an aqueous solution of sodium
Answer:
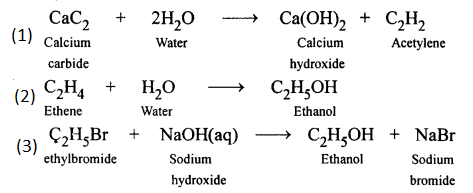
Question 4.
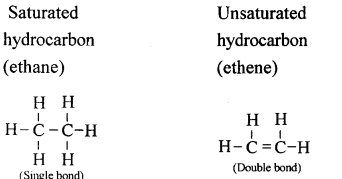
Distinguish between the saturated hydrocarbon ethane and the unsaturated hydrocarbon ethene by drawing their structural formulae.
Answer:
Question 5.
Which type of reaction i.e. addition or substitution is shown by ethane and ethene ?
Answer:
ethane→ Substitution, ethene → addition reaction
Question 6.
Write the equation for the complete combustion of ethane.
Answer:
2C2H6 + 7O2 → 4CO2 + 6H2O Δ
Question 7.
Name the alcohol, aldehyde and acid formed when ethane is oxidised.
Answer:
Alcohol obtained from ethane is ethyl alcohol [C2H5OH]. The aldehyde obtained from ethane is acetaldehyde [CH3 CHO]. The acid obtained from ethane is acetic acid [CH3COOH].
Question 8.
Why is pure acetic acid known as glacial acetic acid ?
Answer:
Pure acetic acid is known as glacial acetic acid because it freezes below 16.5°C to an icy mass (glacier).
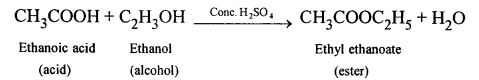
Question 9.
What type of compound is formed by the reaction between acetic acid and an alcohol ?
Answer:
Ester is formed by the reaction between acid and an alcohol.
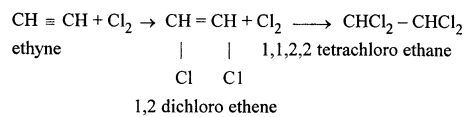
Question 10.
By what type of reaction could a compound containing C, H and Cl – be obtained from ethyne ?
Answer:
Addition reaction
Question 11.
State the term for the reaction in which the hydrogen of an alkane is replaced by chlorine.
Answer:
Substitution reaction.
2009
Question 1.
Which of the following statements is wrong about lkanes ?
(A) They are all saturated hydrocarbons.
(B) They can undergo addition as well as substitution reaction.
(C) They are almost non polar in nature.
(D) On complete combustion give out carbon dioxide and water.
Question 2.
Write balanced equation for : Acetic acid is warmed with ethanol in the presence of con. H2SO4.
Answer:
![]()
Question 3.
Find the odd one out in each case and explain your choice.
- C3H8 ,C5H10, C2H6, CH4
- Formic acid, Nitric acid, Acetic acid, Propanoic acid.
Answer:
- C5H10 [All others are alkane, this is an alkene]
- Nitric acid [This is the only inorganic acid rest all are organic acids]
Question 4.
Identity the substances ‘S’ based on the information given below:
The reddish brown liquid ‘S’ is dissolved in water. When ethyne gas is passed through it, ttirns colourless.
Answer:
Bromine solution
Question 5.
Fill in the blanks with the correct words from the brackets.
Generally ionic compounds exist in (i) ………………. (solid/liquid/gas) state. Melting and boiling points of covalent compounds are generally (ii) …………….. (low/high). The general formula for alkane is (iii)……………. (CnH 2n / Cn H 2n-2 t/Cn H2n+2). For alkynes the geperal formula is (iv)…… (CnH2n / CnH2n-2/CnH2n+2)
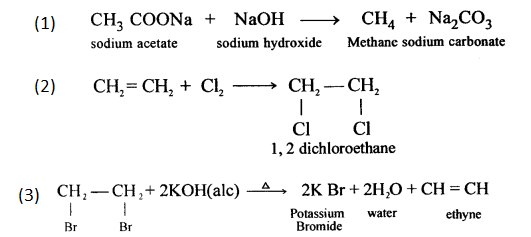
Question 6.
Give chemical equation for the following :
- The laboratory preparation of methane from sodium acetate.
- The reaction of one mole of ethene with one mole of chlorine gas.
- The preparation of ethyne from 1, 2 – dibromoethane.
Answer:
Question 7.
State how the following conversions can be carried out:
- Ethyl chloride to Ethyl alcohol.
- Ethyl chloride to Ethene.
- Ethene to Ethyl alcohol.
- Ethyl alcohol to Ethene.
Answer:
- By treating ethyl chloride with aqueous KOH.
- By heating ethyl chloride with alcoholic KOH.
- By passing ethene into concentrated H2SO4 at 80°C and high pressure or by hydrating of ethene.
- By heating ethyl alcohol with concentrated H2SO4 at 170°C.
Question 8.
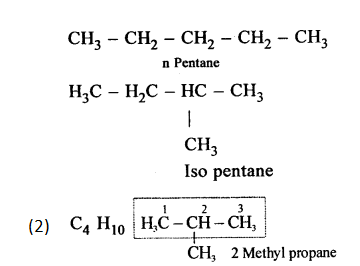
Define isomerism. Give the IUPAC name of the isomer C4H10 which has a branched chain.
Answer:
(1) Isomerism : Organic compounds having the same formula but different structural formulae are called isomers and this property is known as isomerism.
Example : n pentane & isopentane
2010
Question 1.
Select the correct answer
- The organic compound, which gives a red precipitate with ammoniacal cuprous chloride and undergoes an addition reaction –
(A) Ethane
(B) Ethene
(C) Ethyne
(D) Ethanol - The organic compound which when mixed with ethyle alcohol, [ethanol], makes it spurious.
(A) Methanol
(B) Methanoic acid
(C) Methanal
(D) Ethanoic acid
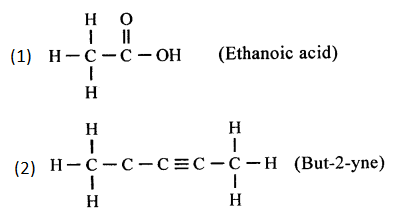
Question 2.
Draw the structural formula of—
1. Ethanoic acid
2. But-2-yne
Answer:
Question 3.
Compound ‘X’ is bubbled through bromine dissolved in CCl4 and the product formed is CH2Br – CH2Br.
- Draw the structural of X and state what type of reaction X has undergone.
- State your observation for the above reaction.
- Name the compound formed when steam reacts with A in the presence of phosphoric acid.
- What is the procedure for converting the product of (b) (iii) back to X ?
Answer:
- CH2Br – CH2Br
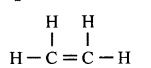
The above reaction is called addition reaction, - The colour of bromine colour fades.
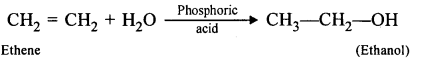
- Ethanol can be converted into ethene, by dehydrating it with cone, sulphuric acid.
2011
Question 1.
Name a gaseous hydrocarbon commonly used for welding purposes.
Answer:
Acetylene
Question 2.
Give reasons for the following –
- almost 90% of all known compounds are organic in nature.
- it is dangerous to burn methane in an insufficient supply of air.
Answer:
- Because of ability of carbon to catenate i.e forms straight chain, branched chains or ring like compounds.
- Because carbon monoxide is produced in an insufficient supply of air. This gas is extremely poisonous for human beings as it cuts off the oxygen supply by forming carboxy haemoglobin in the blood.
Question 3.
Choose the correct answer –
- The functional group present in acetic acid is:
(A) Ketonic C = O
(B) Hydroxyl-OH
(C) Aldehydic – CHO
(D) Carboxyl – COOH - Unsaturated hydrocarbons undergo :
(A) a substitution reaction
(B) an oxidation reaction
(C) an addition reaction
(D) none of the above - The number of C – H bonds in ethane molecule are:
(A) Four
(B) Six
(C) Eight
(D) Ten
Question 4.
Select the correct answer the choices given :
- The catalyst used for conversion of ethene to ethane is commonly……… [nickel/iron/cobalt]
Ans: Nickel - Acetaldehyde when oxidized with acidified potassium dichromate, forms ……… [ester/ethanol/
acetic acid]
Ans: Acetic acid - Ethanoic acid reacts with ethanol in presence of cone. H2SO4, so as to form a compound and water. The chemical reaction which takes place is called…………[dehydration/hydrogenation/esterification]
Ans: Esterification
Question 5.
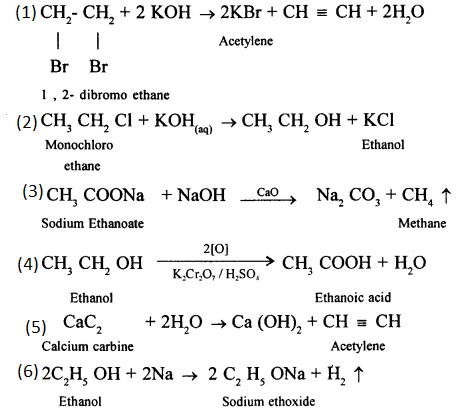
Write balanced chemical equations for the following :
- Write the equation for the reaction taking place between 1, 2 – dibromoethane and ajcoholic potassium hydroxide.
- Monochloro ethan is hydrolysed with aqueous KOH.
- A mixture of sodalime and sodium acetate is heated.
- Ethanol under high pressure and low temperature is treated with acidified potassium dichromate.
- Water is added to calcium carbide.
- Ethanol reacts with sodium at room temperature.
Answer:
2012
Question 1.
State the observation : Bromine vapours are passed into a soln. of ethyne in carbon tetrachloride.
Answer:
Ethyne decolorizes the reddish brown colour of bromine solution.
Question 2.
From – Ethyne, ethanol, acetic acid, ethene, methane. Choose the one which relates to (i) to (iv).
- An unsaturated hydrocarbon used for welding purposes.
Ans: Ethyne - An organic compound whose functional group is carboxyl.
Ans: Acetic acid - A hydrocarbon which on catalytic hydrogennation gives a saturated hydrocarbon.
Ans: Ethene - An organic compound used as a thermometric liquid.
Ans: Ethanol
Question 3.
(1) Why is pure acetic acid known as glacial acetic acid?
(2) Give a chemical equation for the reaction between ethyl alcohol and acetic acid.
Answer:
(1) Pure acetic acid freezes below 20°C to form a transparent solid which looks like ice and hence, it is called glacial acetic acid.
(2) ![]()
Question 4.
Rewrite the correct statement with the missing word/s. Ethyl alcohol is dehydrated by sulphuric acid at a temperature of about 170°C.
Answer:
Ethyl alcohol is dehydrated by concentrated sulphuric acid at a temperature of about 170°C.
Question 5.
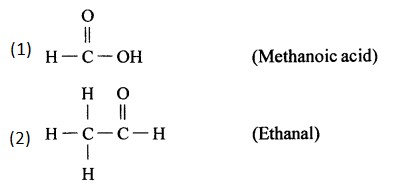
Give the structural formula for the following :
- Methanoic acid
- Ethanal
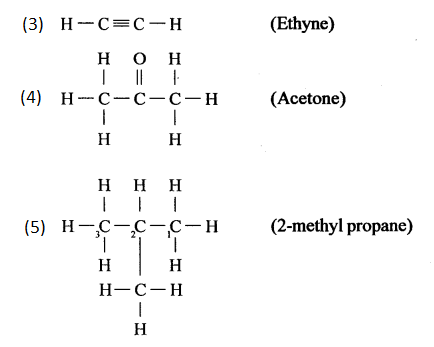
- Ethyne
- Acetone
- 2-methyl propane.
Answer:
2013
Question 1.
Identify the gas evolved when : sodium propionate is heated with soda lime.
Answer:
Ethane gas
Question 2.
Give suitable chemical term for : A reaction in which hydrogen of an alkane is replaced by a halogen.
Answer:
Subsitution reaction
Question 3.
Give a chemical test to distinguish between : Ethene gas and ethane gas.
Answer:
To the given gas add few drops of bromine solution in carbon, tetra-chloride. In case of ethene gas, the reddish colour of bromine discharges. However, in case of ethane gas the reddish colour of bromine does not discharge.
Question 4.
Identify the statement that is incorrect about alkanes :
(A) They are hydrocarbons.
(B) There is single covalent bond between carbon and hydrogen
(C) They can undergo both substitution as well as addition reactions
(D) On complete combustion they produce carbon dioxide and water.
Question 5.
Give balanced equations for the laboratory preparations of:
- A saturated hydrocarbon from iodomethane.
- An unsaturated hydrocarbon from an alcohol.
- An unsaturated hydrocarbon from calcium carbide.
- An alcohol from ethyl bromide.
Answer:
- CH3I + 2H (from Zn/Cu couple) → CH4 + HI
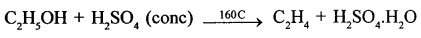
- CaC2 + 2H2O → Ca(OH)2 + C2H2
- C2H5Br + KOH (aq) → C2H5OH + KBr
Question 6.
Give the structural formulae for :
- An isomer of n-butane.
- 2-propanol.
- Diethyl ether.
Answer:
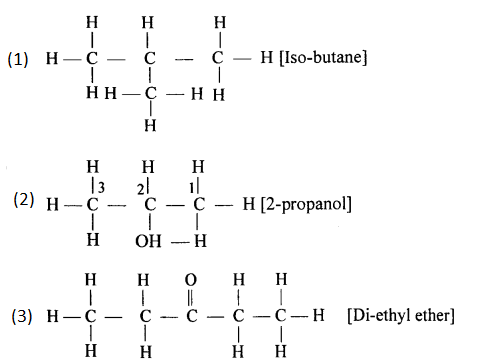
Question 7.
Give reasons for :
- Methane does not undergo addition reactions, but ethene does.
- Ethyne is more reactive than ethane.
- Hydrocarbons are excellent fuels.
Answer:
- All the four covalent bonds between the carbon and hydrogen are fully shared. Thus the hydrogen atom can only be substituted by more reactive atoms or group of atoms. There is no scope of addition of reactive atoms in its molecule.
However, in case of ethene there is a double bond between the two carbon atoms. These bonds are under strain and hence can be easily broken by more reactive atoms to form addition compounds which are saturated in nature. - Ethyne has a triple covalent bond (—C = C —) between two carbon atoms, whereas ethene has a double covalent bond (—C = C —) between the two carbon atoms. So, the strain in the bounding of ethyne is far more than ethene. This accounts of the reactivity of ethyne as its bonds break more easily than that of ethene.
- All the constituents of hydrocarbon (carbon and hydrogen) are highly combustible and do not have any uncombustible content. So, hydrocarbons are excellent fuels.
2014
Question 1.
The I.U.P.A.C. name of acetylene is,
(A) propane
(B) propyne
(C) ethene
(D) ethyne.
Question 2.
Ethanol reacts with sodium to give………(sodium ethanoate, sodium ethoxide, sodium propanoate)
Answer:
sodium ethoxide
Question 3.
Give one word or phrase for – hydrocarbons containing a
![]() functional group
functional group
Answer:
Hydrocarbons containing a![]() functional group → Alkan one or Ketonic functional group.
functional group → Alkan one or Ketonic functional group.
Question 4.
Write balanced equation for preparation of
- ethane from sodium propionate.
- ethanol from monochloroethane and aq. sodium hydroxide.
Answer:
- Preparation of ethane from sodium propionate.
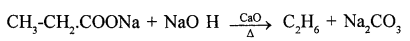
- Preparation of ethanol from monochloroethane and aq. sodium hydroxide.
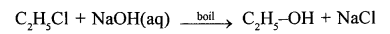
Question 5.
Distinguish between : Ethane and ethene (using alkaline potassium permanganate solution)
Answer:
Ethane and ethene (using alkaline KMnO4)
Ethene decolourises the colour of alkaline KMnO4 but ethane does not
Question 6.
State the conditions required for :
- Catalytic hydrogenation of ethyne.
- Preparation of ethyne from ethylene dibromide.
Answer:
(1) One volume of ethyne gas is mixed with two volumes of hydrogen gas and passed over heated nickel at 300°C, when an addition reaction takes place with the formation of ethane gas.
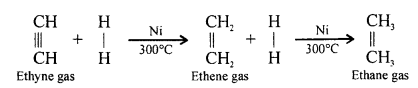
(2) Zinc dust is added to a mixture of 95% ethyl alcohol and 5% of ethylene dibromide. The mixture is gently warmed when ethyne gas is liberated.
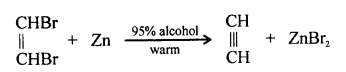
Question 7.
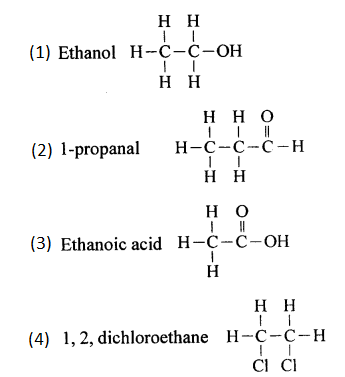
Write structural formula of:
(1) Ethanol
(2) 1-propanal.
(3) ethanoic acid.
(4) 1, 2, dichloroethane.
Answer:
Give the structural formula:
Question 8.
Match A and B with (i) and (ii) :
A: alkynes (1) CnH2n+2
B: alkane (2) CnH2n-2
Answer:
A: alkynes (2) CnH2n-2
B: alkane (1) CnH2n+2
2015
Question 1.
Select from the list — Ammonia, ethane, hydrogen chloride, hydrogen sulphide, ethyne
- The gas is used for welding purposes.
Ans: Ethyne - This gas is also a saturated hydrocarbon.
Ans: Ethane
Question 2.
State which of the following statements does not describe the property of alkenes :
(A) They are unsaturated hydrocarbons
(B) They decolourise bromine water
(C) They can undergo addition as well as substitution reactions
(D) They undergo combustion with oxygen forming carbon dioxide and water.
Ans. (C) They can undergo addition and substitution reactions. Alkenes do not undergo substitution reaction.
Question 3.
State one appropriate observation when : The gaseous product obtained by dehydration of ethyl alcohol is passed through bromine water.
Answer:
The reddish brown colour of bromine solution gets decolourised.
Question 4.
Give balanced chemical equations for the following conversions:
- Ethanoic acid to ethyl ethanoate.
- Calcium carbide to ethyne.
- Sodium ethanoate to methane.
Answer:
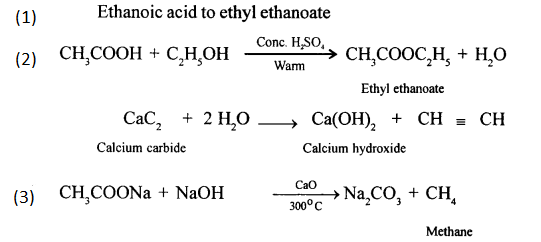
Question 5.
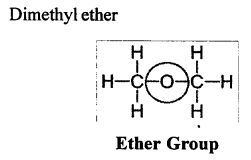
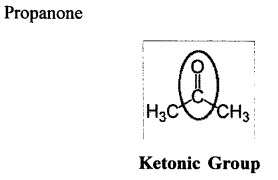
Using their structural formulae identify the functional group by circling them:
(1) Dimethyl ether.
(2) Propanone
Answer:
Dimethyl ether.
Propanone
Question 6.
Name the following :
- Process by which ethane is obtained from ethene.
- A hydrocarbon which contributes towards the greenhouse
- Distinctive reaction that takes place when ethanol is treated with acetic acid.
- The property of element by virtue of which atoms of the element can link to each other in the form of a long chain or ring structure.
- Reaction when an alkyl halide is treated with alcoholic potassium hydroxide.
Answer:
- Hydrogenation (addition)
- Methane
- Esterification
- Catenation
- Dehydrohalogenation
2016
Question 1.
Fill in the blanks : Conversion of ethene to ethane is an example of………… (hydration / hydrogenation).
Answer:
Conversion of ethene to ethane is an example of hydrogenation.
Question 2.
Write balanced chemical equations for : Preparation of ethanol from ethyl chloride.
Answer:
C2H5C1 + NaOH(aq) C2H5OH + NaCl
Question 3.
Identify the term/substance in each of the following :
- The catalyst used in the conversion of ethyne to ethane.
- The type of reactions alkenes undergo.
Answer:
- Nickel or platinum or palladium.
- Addition reactions.
Question 4.
Write the IUPAC names of:
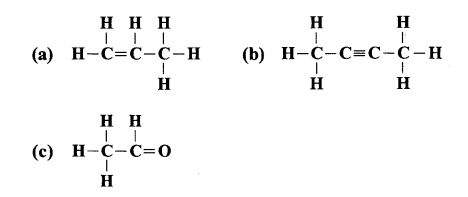
Answer:
(a) Propene
(b) 2-butyne
(c) ethanal
Question 5.
Write a balanced chemical for :
- Burning of ethane in plentiful supply of air.
- Action of water on calcium carbide.
- Heating of Ethanol at 170°C in the presence of cone, sulphuric acid.
Answer:
(1) C2H6 + 7/2 O2 → 2CO2 + 3H2O
2C2H6 + 7O2 → 4CO2 + 6H2O
(2) CaC2 + 2H2O → Ca(OH)2 + C2H2↑
(3) 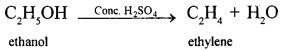
Question 6.
Give the structural formulae of:
(1) 2-methyl propane
(2) Ethanoic acid
(3) Butan – 2 – ol
Answer:
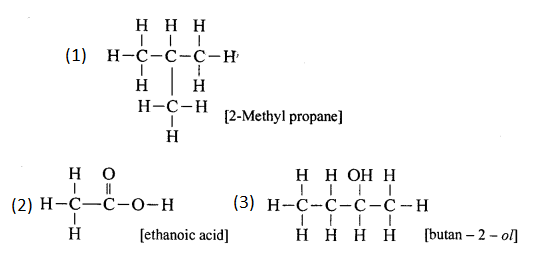
Question 7.
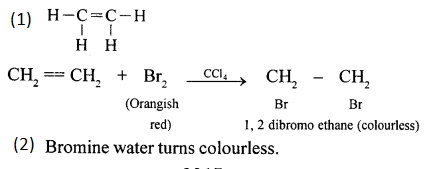
Compound A is bubbled through bromine dissolved in carbon tetrachioride is as follows :
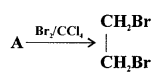
(1) Draw the structure if A.
(2) State your observation during this reaction.
Answer:
2017
Question 1.
Fill in the blanks from the choices given in brackets – The compound formed when ethene reacts with hydrogen is……….. [CH4, C2H6, C3H8]
Answer:
The compound formed when ethene reacts with hydrogen is C2H6.
Question 2.
Choose the correct answer from the options given – If the molecular formula of an organic compound is C10H18 it is –
(A) Alkene
(B) Alkane
(C) Alkyne
(D) Not a hydrocarbon
Question 3..
Identify the substance underlined – An organic compound containing – COOH functional group.
Answer:
Ethanoic acid [CH3—COOH]
Question 4.
Write the balanced chemical equation for – Preparation of methane from iodomethane
![]()
Question 5.
Identify the term or substance based on the descriptions given below:
- Ice like crystals formed on cooling an organic acid sufficiently.
- Hydrocarbon containing a triple bond used for welding purpose.
- The property by virtue of which the compound has the same molecular formula but different structural formulae.
- The compound formed where two alkyl groups are linked by
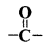 group.
group.
Answer:
- Glacial acetic acid
- Ethyne or acetylene
- Isomerism
- Ketone or Alkanone
Question 6.
Give a balanced chemical equation for each of the following –
- Preparation of ethane from sodium propionate.
- Action of alcoholic KOH on bromoethane.
Ans:
- C2H5COONa + NaOH → C2H6 + Na2CO3
- CH3Br + KOH → CH3OH + KBr
Question 7.
State one relevant observation for the following reaction – Addition of ethyl alcohol to acetic acid in the presence of concentrated sulphuric acid.
Answer:
On warming the mixture gives fruity smell.
Question 8.
Draw’ the structure formula for each of the following –
- 2, 3 – dimethyl butane
- Diethyl ether
- Propanoic acid
Answer:
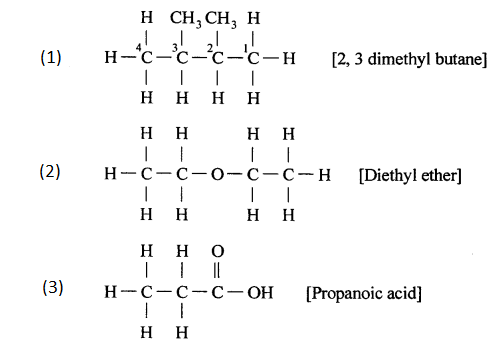
ADDITIONAL QUESTIONS
Question 1.
Explain the term ‘Organic Chemistry ’. State the ‘Natural sources ’ and ‘Importance’ of organic compounds.
Answer:
- Organic Chemistry-It is the chemistry of specific carbon compounds except – oxides, carbonates, bicarbonates and metallic carbides.
- Plants, Animals, Petroleum, dyes and drugs are all natural sources.
- Compounds of organic origin are : Food – carbohydrates, vitamins Dyes-azodyes Clothing – cotton, silk and wool Fuels – petrol Medicines – penicillin Explosives – trinitrotoluene.
Question 2.
Explain the ‘unique nature of carbon atom’ with specific reference and meaning to —
(a) ‘Tetravalency’ — leading to formation of single, double and triple bonds
(b) ‘Catenation’ — leading to formation of straight chain, branch chain and cyclic compounds.
Answer:
Some unique properties shown by carbon atom are :
(a) Tetravalency
(b) Catenation
(c) Ability to form multiple bonds.
(a) Tetravalency : Atomic number of carbon is 6. Its electronic configuration is 2, 4. Therefore, it has four electrons in its valence shell. Carbon atom can neither lose nor gain electrons to complete its octet (not possible from energy point of view). Therefore, carbon atom completes its octet by sharing four electrons with other atoms, i.e., it can form four covalent bonds, called its tetracovalency.
For example:
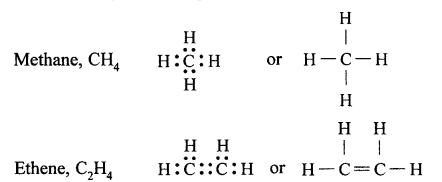
(b) Catenation: The property by virtue of which a large number of atoms of the same element get linked together through single or multiple covalent bonds, forming straight or branched chains and rings of different sizes, is called catenation. Carbon shows catenation to the maximum extent due to strong carbon-carbon bonds and its tetracovalency.
In this process of catenation, carbon atoms form straight or branched chains and cyclic rings of various sizes and can involve single, double or triple covalent bonds.
Question 3.
State reasons for ‘Justification of a separate branch’ for ‘Organic Chemistry.
Answer:
This is due to the following reasons:
- The number of known organic compounds is very large as compared to the number of known inorganic compounds.
- Organic compounds involve only a few elements (C, H, O, N, S, P, F, Cl, Br, I etc.), whereas inorganic compounds involve all the known elements.
- Organic compounds have complex nature and have high molecular mass.
- Organic compounds involve covalent bonds whereas inorganic compounds involve electrovalent bonds.
- Organic compounds show isomerism whereas inorganic compounds do not show isomerism.
- The properties of organic compounds are different from inorganic compounds.
All these facts convince us to study organic chemistry as a separate branch of chemistry.
Question 4.
State five differences between the characteristics of organic and inorganic compounds. State how organic compounds are classified.
Answer:
(a) Characteristics of organic compounds :
- These are made up of only a few elements C, H, O, N, S,X(C1, Br,l)
- These involve covalent bonds.
- These are generally gases or liquids
- They have low melting and boiling points.
- They are combustible.
- They show molecular reations.
- They show isomerism.
- These are non-conductors of electrocity.
- There are generally insoluble in water but soluble in organic solvents.
Characteristics of inorganic compounds :
- These are made up of all the known elements.
- These involve ionic bonds.
- These are generally solids.
- They have high melting and boiling points.
- They are non-combustible.
- They show ionic reactions.
- They don’t show isomerism.
- These are generally good conductors of electricity.
- These are generally soluble in water but insoluble in organic solvents.
(b) Classification of Organic Compounds Aliphatic – Open Chain Compounds

Question 5.
Explain the term ‘Homologous series’. State the general characteristics of members of the series with special reference to molecular mass or molecular formula.
Answer:
Homologous series is a series of organic compounds, that are grouped into a smaller number of series of compound.
General Characteristics of homologous series :
- The members of a series have same functional group.
- Two consecutive members of a homologous series differs each other in their composition
by – CH2unit
Example :
Alcohol (-OH)
CH3 – OH, CH3 – CH2 – OH, CH3 – CH2 – CH2 – OH - The members of a homologous series can be represented by same general formula.
Example :
Alcohol-CnH2n+1OH
Aldehyde — CnH2n+1|CHO
Carboyxlic acid — CnH2n+1COOH - The members of a particular homologous series have almost same chemical properties due to presence of same functional group.
- The physical properties (like solubility, melting point, boiling point, state) of members of a homologous series either gradually increase or decrease with increase in molecular mass.
- The members of a homologous series can be prepared by same or common general method of preparation.
- The first member of homologous series generally shows certain different chemical behavior than other members of the series.
Question 6.
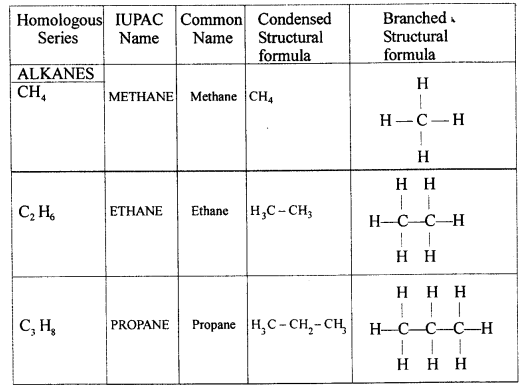
Differentiate between — ‘Molecular formula’ and ‘Structural formula’ — of an organic compound. Write the ‘condensed structural formula and ‘branched structural formula’ of ethene.
Answer:
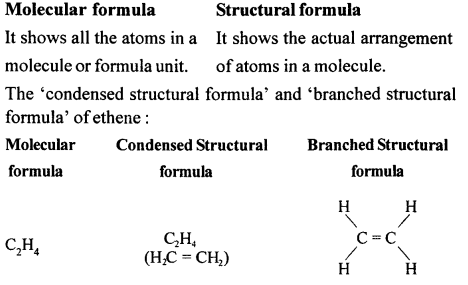
Question 7.
State what are ‘Alkyl groups ’. State the alkyl group of the parent alkane — methane and ethane.
Answer:
Alkyl Group : It is obtained by removing one hydrogen atom from a molecule of an alkane.
Methane : Methyl (Alkyl group)
Ethane : Ethyl (Alkyl group)
Question 8.
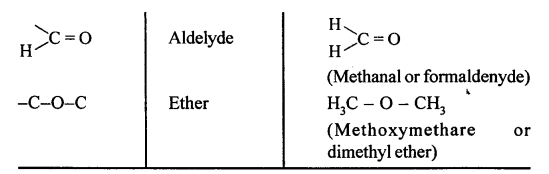
State what are ‘Functional groups’. Name the following functional groups —
![]()
X = -F, -Cl, -Br, -I ; -C=O; -C-O-C
Answer:
Functional Groups : An atom, radical or bond which defines the structure of an organic compound and give if its characteristic properties
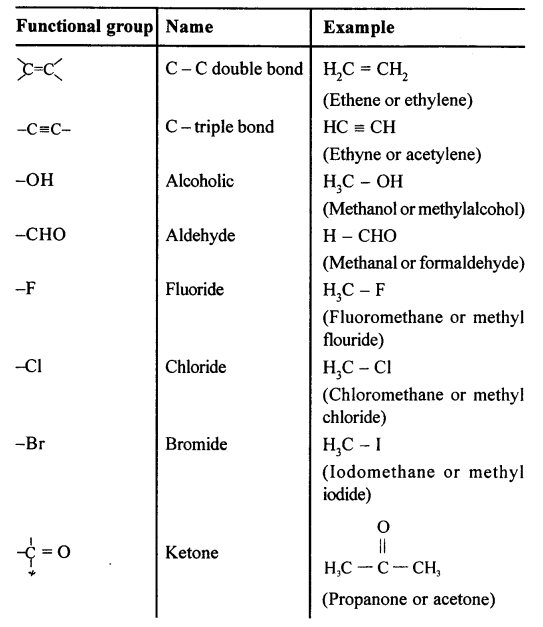
Question 9.
Explain the terms — ‘Isomers’ and ‘Isomerism’. State the ‘Characteristics of isomers’ with reference to —
Properties of isomers ; Number of isomers with relation to carbon atoms in the isomer.
Differentiate between — ‘Chain isomerism’ and ‘Position isomerism ’ – with suitable examples.
Answer:
Two or more compounds having the same molecular formula but different physical and chemical properties are called isomers and this phenomenon is known as isomerism.
Isomers have the same number of atoms of each element in them and the same atomic weight but differ in other properties. For example, there are two compounds with the molecular formula C2H6O. One is ethanol (ethyl alcohol), CH3CH2OH, a colorless liquid alcohol; the other is dimethyl ether, CH3OCH3, a colorless gaseous ether.
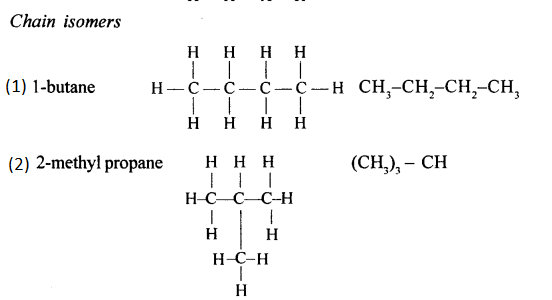
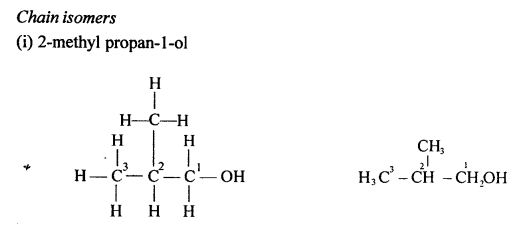
Alkanes with more than three carbon atoms form isomers. The various isomers differ in the framework of the carbon chains.
Differentiate between — ‘Chain isomerism’ and ‘Position isomerism’
Chain isomers: Compounds having same molecular formula with difference in carbon chain pattern like linear or branch are called chain isomers. 1-Pentyne is chain isomer for 3-methyl Butyne.
CH3 – CH2 – CH2 – C = CH and CH3– C(CH3) – C = CH
Position isomers: Compounds having same molecular formula ^ with difference in position of the functional group are called position isomers. 1-Butyne and 2-Butyne are position isomers.
CH3– CH2– C=CH and CH3 – C = C – CH3.
Question 10.
Explain the term – ‘Nomenclature’. State its need with reference to organic compounds. State the basic rules of Nomenclature by the trivial system – with suitable examples. Explain the longest chain rule and the smallest number for functional groups rule of Nomenclature by the IUPAC system – with suitable examples.
Answer:
(a) Nomenclature :
Nomenclature is the system of assignment of names to organic compounds.
Need for Nomenclature : Very large number of organic compounds with varying molecular structure need a systematic method of nomenclature. Further many a times same molecular formula represents two or more compounds (isomerism).
(b) Nomenclature by Trivial System:
In this method, name of an organic compound is derived from its;
- Source (e.g., benzoic acid is obtained by distillation from gum benzoin, fructose or fruit sugar from fruits etc).
- Latin or Greek origin (e.g., formic acid, HCOOH is present in sting of red ants, formicus in Latin means an ant).
- Properties (e.g., palmitic acid is an acid derived from palm oil etc).
(c) Longest Chain Rule :
- In the nomenclature of alkanes, the longest continuous chain if C-atoms is selected. For this, alkyl groups, if present, are written in the expanded form.
For example,
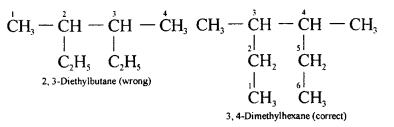
- Smallest Number for Substituent : Once the principal chain is selected, it is numbered in such a say that the substituent gets the lowest number.
For example,
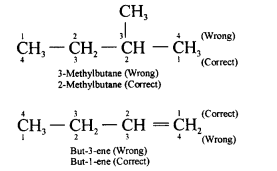
Question 11.
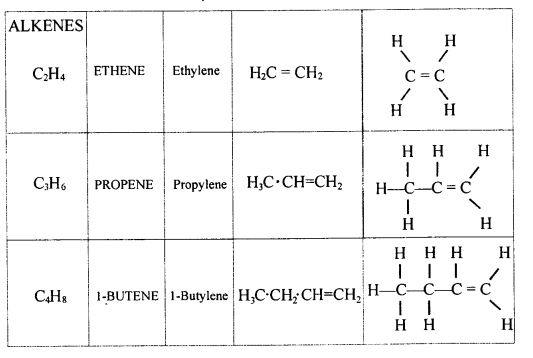
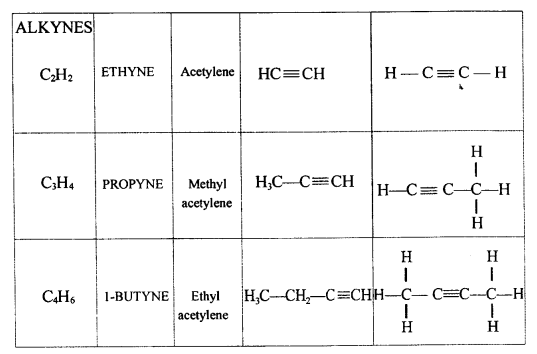
Explain the term – ‘Hydrocarbons’. State the two main groups of hydrocarbons with examples. Draw a chart differentiating — ‘Alkanes, Alkenes andAlkynes’ — with respect to:
- General formula
- Characteristic bond
- IUPAC and the common name of the first three members and condensed/branched/electronic structural formula of each
- Availability of electrons
- Reactivity
- Characteristic reaction.
Answer:
Hydrocarbons — They are aliphatic open chain organic compounds containing carbon and hydrogen only.
Molecular formula is CxHy where X and Y are whole numbers.
- Saturated hydrocarbons — Homologous series of alkanes.
- Unsaturated hydrocarbons — Series of alkynes and alkenes.
- General formula :
Alkanes — CnH2n+2
Alkenes — CnH2n
Alkynes — CnH2n_2
- Characteristic bond
Alkanes → C – C < single bond
Alkenes → C = C < double bond
Alkynes → C = C < triple bond - IUPAC Name Condensed/branched/electronic structural formula of each
 ..
..
- Availability of electrons
Alkanes — Not available
Alkenes — Available Alkynes — Available - Reactivity
Alkanes — Less reactive
Alkenes — More reactive
Alkynes — Most reactive - Characteristic Reaction
Alkanes — Substitution reaction
Alkenes — Addition reaction
Alkynes — Addition reaction
- General formula :
Question 12.
Draw the structural formula of each of the following :




Answer:
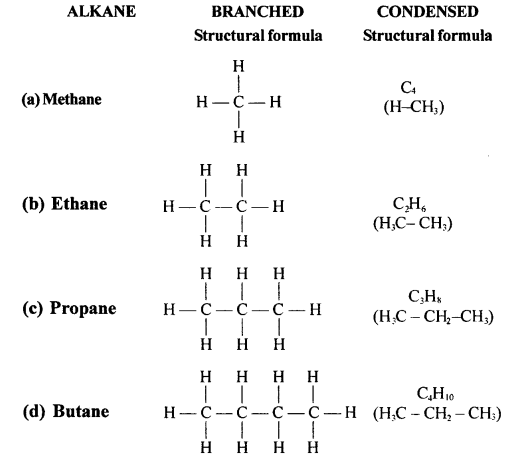
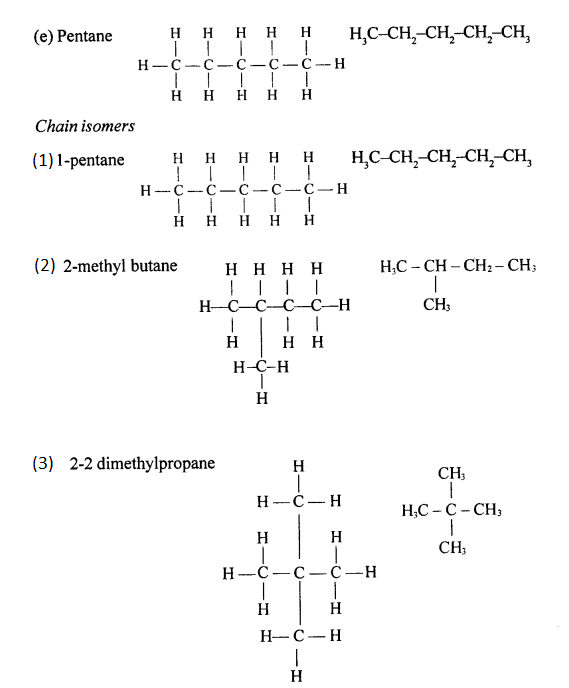
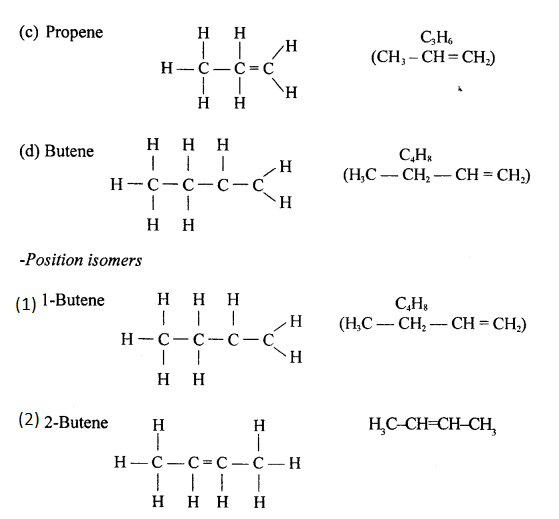
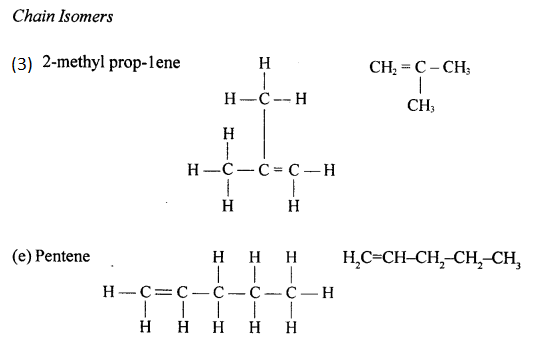
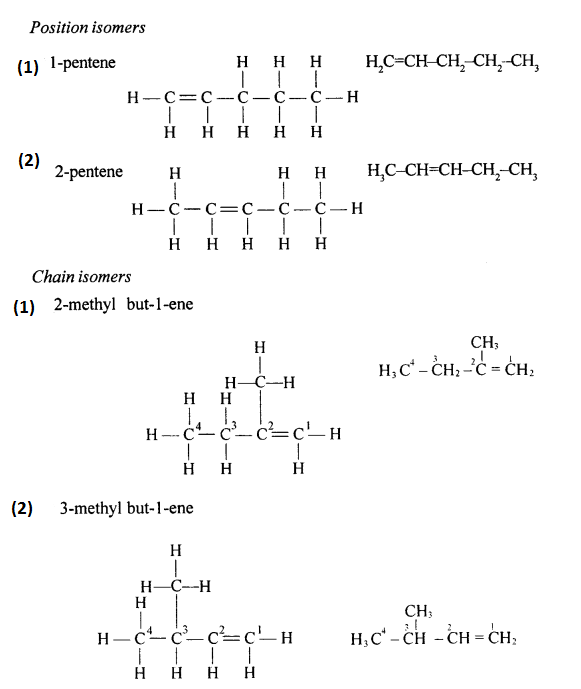
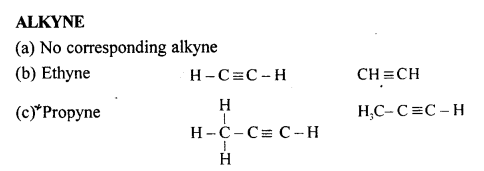
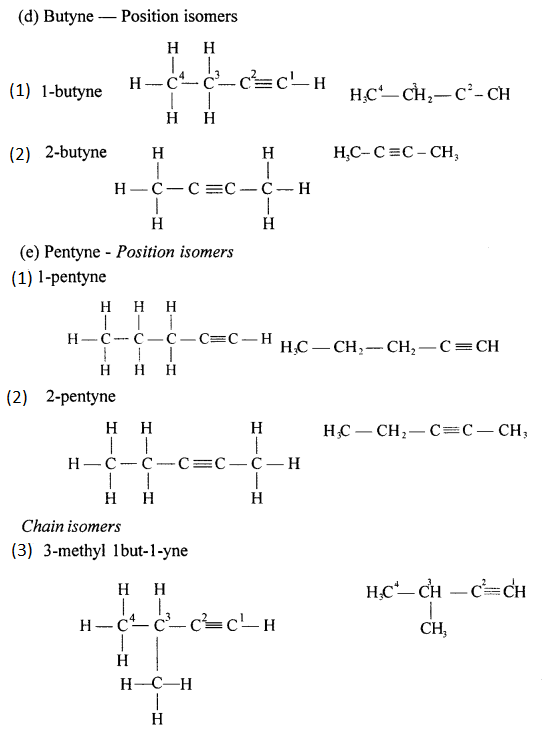
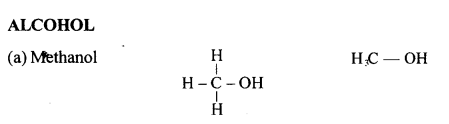
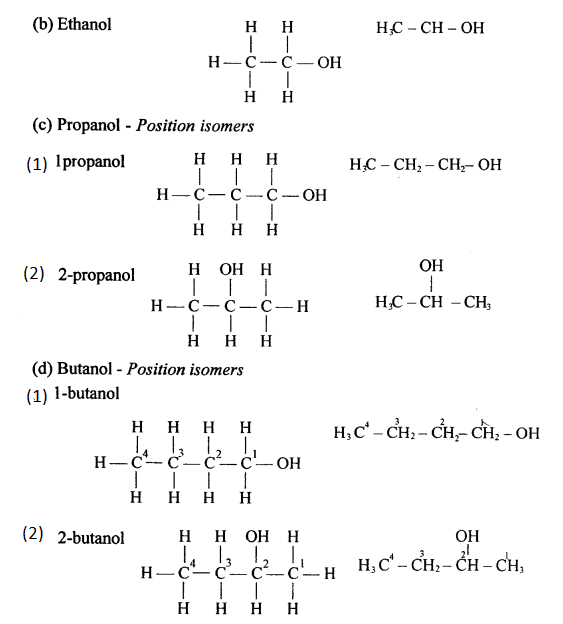
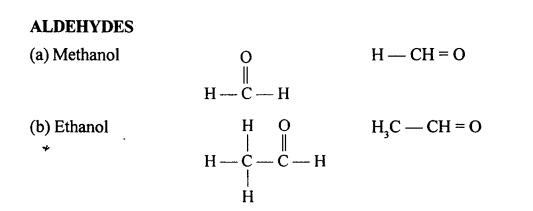
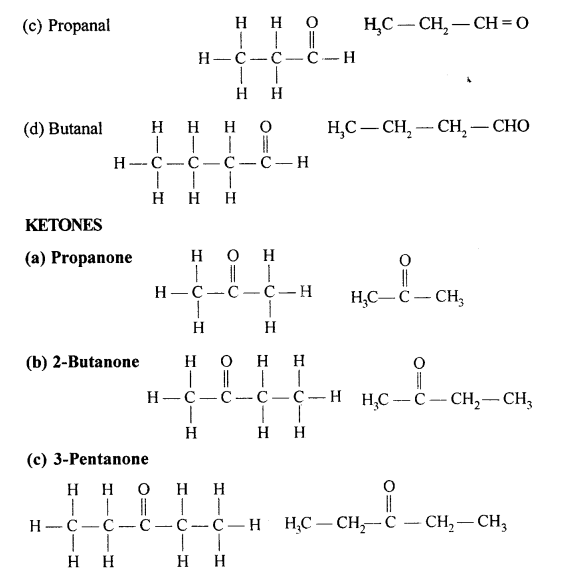
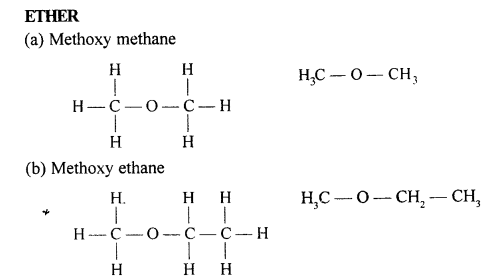
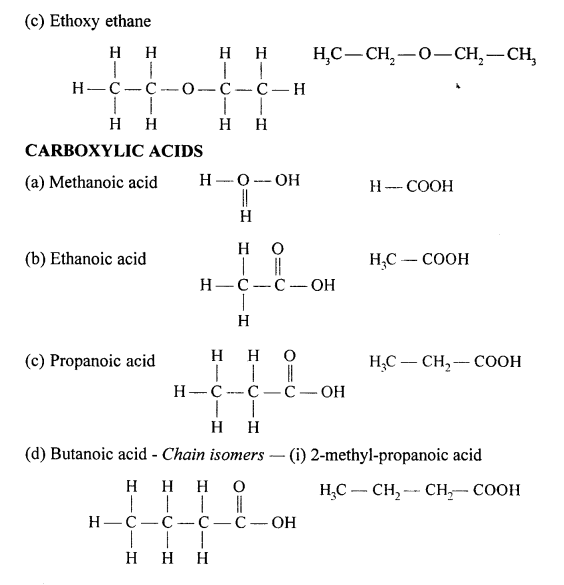
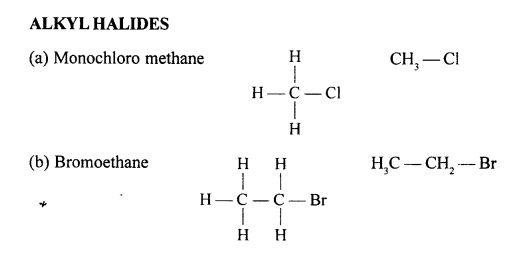
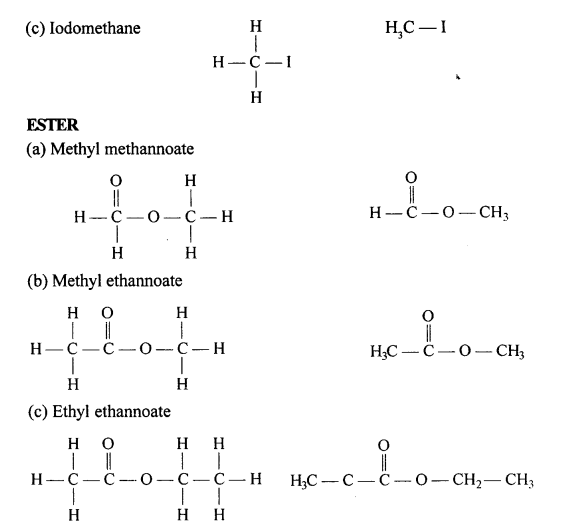
Question 13.
Give the IUPAC name of the compounds numbered (I) to (y).
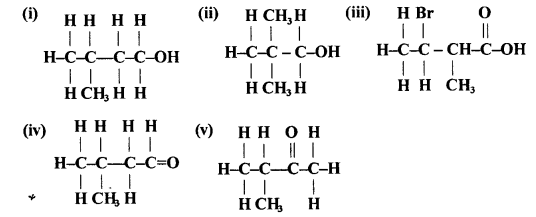
Answer:
- Methyl butanol, 2-Methyl-1-butanol
- 2, 2-dimethyl propanol
- 2-Bromocyclo pentan- I -ol
- 3-Methylbutanal
- 3-Methyl-2-butanone
Question 14.

Answer:
(a) CH3COONa + NaOH → CH4 + Na2CO3
(b) CH3I + 2|H| → CH4 + HI
(c) C2H5COONa + NaOH → C2H6 + NaCO3
(d) C2H5Br + 2(H) → C2H6 + HBr
Question 15.
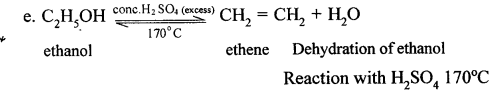
Give equations for the conversions of – Methane, Ethane, Ethene, Ethyne, Methanol, Ethanol and Ethanoic Acid.
Answer:


Answer:
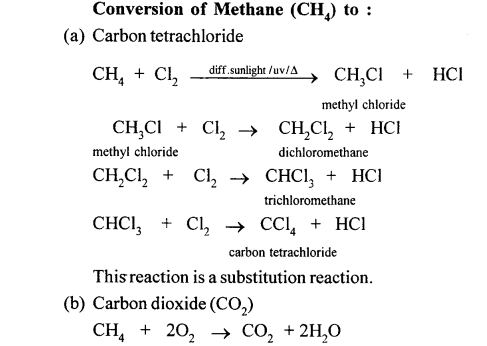
When methane is burnt in excess of air or oxygen with pale blue flame it gives carbon dioxide gas, water and heat energy. This reaction is complete oxidiation reaction.
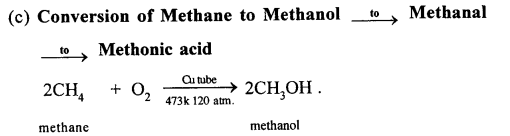
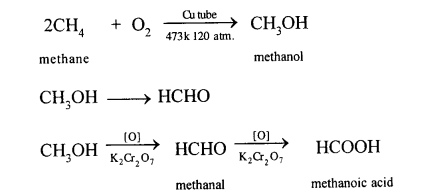
This reaction is carried out in a copper tube. Cu acts as a catalyst. In this reaction, methane is oxidised to methanol or methyl alcohol. In this reaction, methane is heated up to a temp, of 200°C under a pressure of 120 atm.
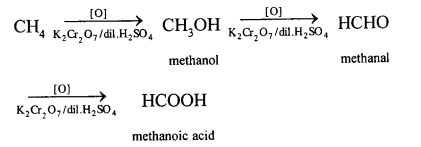
Methane can also be converted into methanol by controlled oxidation of methane in the presence of acidified K2Cr2O7.
In this reaction, methane is oxidised in a copper tube methane is heated upto a tem. of 475 k under a pressure of 120 atm. Copper tube acts as a catalyst. This is carried out as catalystic oxidation.
(d) Conversion of methane to Methanal
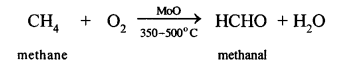
This reaction involves the catalytic oxidation. In this reaction, methane is heated with catalyst molybdenum oxide (MoO) it a temp, of 350 – 500°C, methanol is formed.
(e) Conversion of Methane to Ethyne (C2H2)
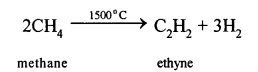
When methane is heated to about 1500°C in an electric arc and then suddenly cooled, the product is C2H2and Hydrogen.
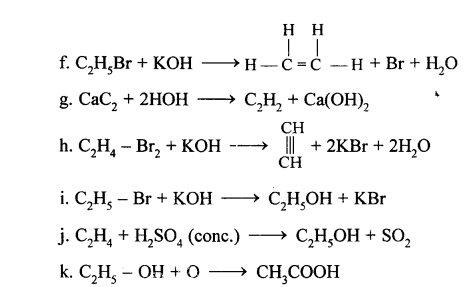
Conversion of Ethane (C2H6) to
(a) Hexachloro ethane (C2Cl6)

C2H4Cl2 + Cl2 → C2H4Cl2 + HCl
C2 H4Cl2 + Cl2→ C2H3Cl3 + HCl
C2 H3Cl3 + Cl2 → C2H2Cl4 + HCl
C2 H2Cl4 + Cl2→ C2HCl5 + HCl
C2HCl5 + Cl2 → C2Cl6 + HCl
This reaction is a substitution reaction.
(b) Carbon dioxide (CO2)
2C2H6 + 7O2 (excess) →4CO2 + 6H2O
![]()

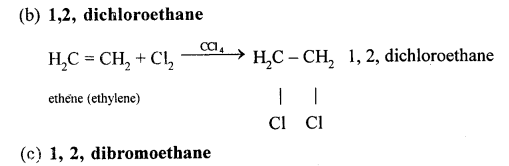
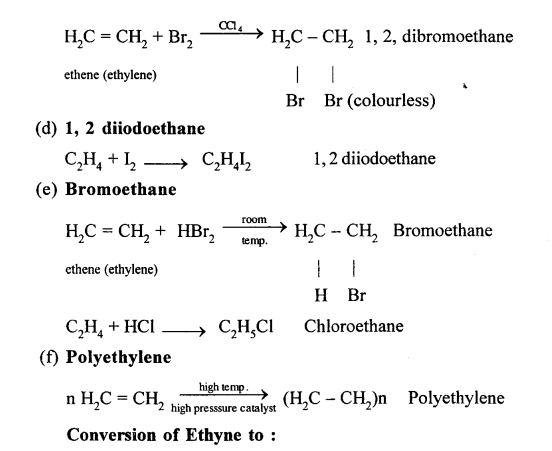
(a) Ethene
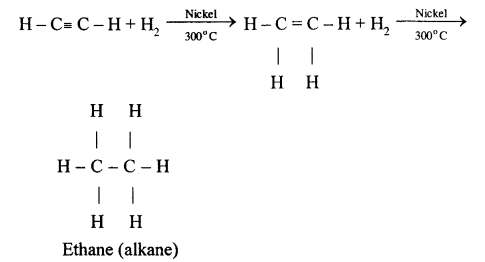
(b) 1,1,2,2, tetrachloroethane
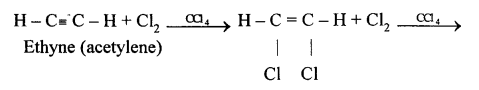

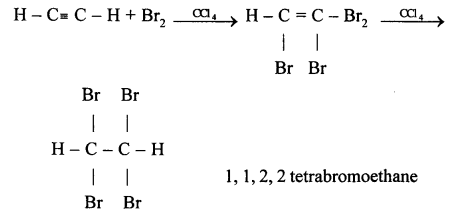
(c) 1,1,2,2 tetrabromoethane
(d) 1,2 diiodoethane
C2H2+I2 →C2H2I2 1,2 diiodoethene
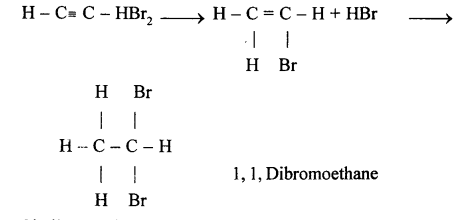
(e) 1,1,Dibromoethane
(f) Copper acetylide / Silver acetylide
HC ≡ CH+ 2CuCl + 2NH2OH →
Cu – C ≡ C – Cu + 2NH2Cl + 2H2O Copper acetylide
HC ≡ CH + 2AgNO3 + 2NH4OH →
Ag – C= C – Ag + 2NH2NO4 + 2H2O Silver acetylide
Conversion of Ethanol to :
(a) Carbon dioxide
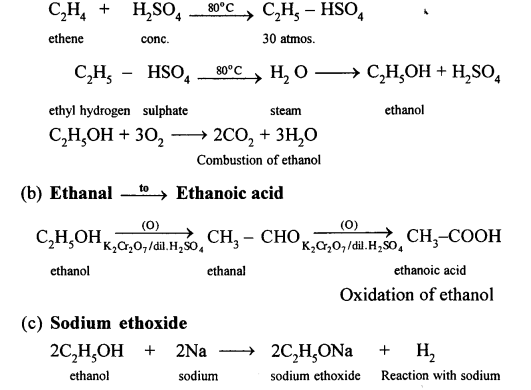
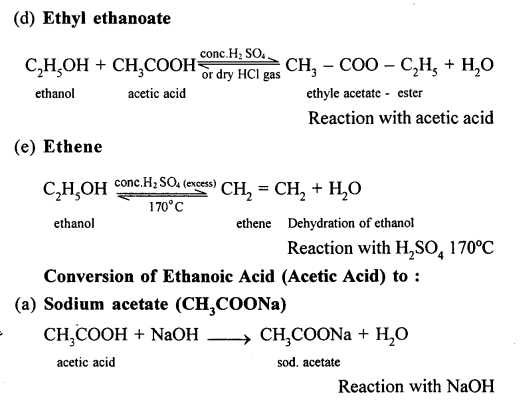
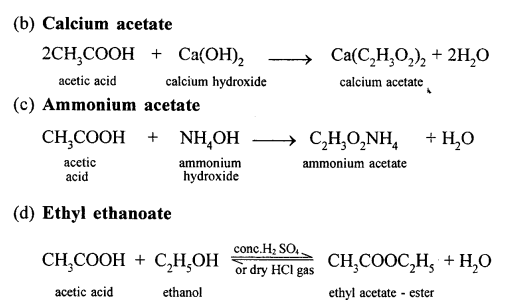
Question 16.
Give reasons for
- alkanes are said to be saturated organic compounds
- alkenes are known as olefins
- alkenes are more reactive than alkanes
- ethanoic acid is known as an aliphatic monocarboxylic acid.
Answer:
- Alkanes do not undergo addition reactions and that is why they are called saturated hydrocarbons or saturated organic compounds. In alkanes all the four valencies of carbon atom are fully satisfied by forming single covalent bonds.
- Alkenes are called olefins because alkenes on treatment with halogens form oily products. (Latin: oleum = oil, ficare = to make)
- Due to the presence of C = C (carbon – carbon double bond) alkenes are more reactive than alkanes.
- Ethanoic acid (CH3 – COOH) contains only one – COOH group (carboxylic acid group) that is why it is called a monocarboxylic acid. As ethanoic acid does not contain a benzene right it is an alphatic monocarboxylic acid.
Question 17.
Explain the terms –
- Denaturated alcohol
- Glacial acetic acid
- Esterification
Answer:
- Denaturated alcohol – Ethyl alcohol containing pyridine or copper sulphate is termed – denaturated alcohol. It is used for – industrial applications only and hence made undrinkable.
- Glacial acetic acid – Anhydrous acetic acid on cooling below 16.5°C crystallizes out in the pure form, forming a crystalline mass resembling ice. Hence pure acetic acid is called glacial acetic acid.
- Esterification – It is known as condensation of an alcohol with an acid. Acetic acid on heating with an alcohol and dehydrating agent [cone. H2SO2] gives an ester – ethyl acetate.
Question 18.
Give a chemical test for to distinguish between
- Ethane, ethene and ethyne
- Ethanol and ethanoic acid.
Answer:
- Tests to distinguish between ethane, ethene and ethyne
- Test — Br2 water test: Pass the gas through Br2 water,
Ethane : Brown colour of Br2 water is not discharged.
Ethene : Brown colour of Br2 water is discharged,
Ethyne : Brown colour of Br2 water is discharged. - Test — Baeyer’s reagent : Pass the gas through
Baeyer’s reagent (alkaline solution of KMnO4).
Ethane : Purple colour of Baeyer’s reagent is not discharged.
Ethene : Purple colour of Baeyers reagent is discharged.
Ethyne : Purple colour of Baeyers reagent is discharged. - Test : Pass the gas through ammoniacal cuprous chloride solution.
Ethane : No ppt.
Ethene : No ppt.
Ethyne : Red ppt. of copper acetylide is formed. - Test : Pass the gas through ammonical silver nitrate solution.
Ethane : No ppt.
Ethene : No ppt.
Ethyne : White ppt. of silver acetylide is formed.
- Test — Br2 water test: Pass the gas through Br2 water,
- Tests to distinguish between ethanol and ethanoic acid
- Test: Add a few drops of blue litmus solution to the given liquid.
Ethanol: No change in colour.
Ethanoic acid : Blue litmus turns red. - Test: Add a pinch of sodium carbonate to the given liquid. Ethanol: No action.
Ethanoic acid : Brisk effervescence with the evolution of C02.
- Test: Add a few drops of blue litmus solution to the given liquid.
Question 19.
Give the main uses of –
- Methane
- Ethane
- Ethene
- Ethyne
- Ethanol
- Ethanoic acid.
Answer:
The main uses of:
(1) Methane and
(2) Ethane –
(a) Illuminant and domestic fuel: In the form of natural gas or gobar gas. [Hydrocarbons – have high calorific value. They are easily combustible and the reaction is exothermic – releasing heat energy. Hence they are excellent fuels]
(b) In manufacture of chemicals : Used as :
- Chloroform : Solvent for rubber, waxes. As an anaesthesia.
- Carbon black : A black pigment in shoe polishes, printers ink etc.
- Formaldehyde : An antiseptic, preservative for biological specimens.
- Methanol : Solvent for varnishes, anti-freeze for automobiles.
- Ethanol: Solvent for resins, a low freezing liquid in thermometers.
(3) Ethene –
(a) Production of oxy-ethylene torch : For welding purposes and cutting metals.
(b) Ripening of green fruits : Artificial ripening and preservation of fruits.
(c) Catalytic hydrogenation: Used in hardening of oils.
(d) It is also used in manufacturing of :
- Synthetic chemicals : Ethylene glycol [anti-freeze], di-ethyl ether [solvent], ethylene oxide [fumigant], mustard gas [chemical warfare],
- Polymers : Polyetheylene, polyvinyl chloride [P.V.C.]- used in packaging, insulators, containers, rain coats etc.
(4) Ethyne –
(a) It is used for producing oxy-acetylene flame for welding and cutting purposes as it produces temperature as high as 3500°C.
(b) It is used as an illuminant in oxyacetylene lamp.
(c) It is used in the manufacture of solvent like westron (C2H2C14) and westrosol (CHCl = CC2).
(5) Ethanol – Main Uses of Ethanol
(a) As a solvent – For gums and resins
(b) In thermometers and spirit levels – Low freezing mobile liquid, [freezing point – 114.1°C].
(c) In manufacture of chemicals – Acetaldehyde [dyes], acetic acid [manufacture of vinegar], chloroform [antiseptic] diethyl ether [anaeshetic].
(6) Ethanoic acid –
Uses :
(a) It is used as a solvent for many organic reactions.
(b) It is used as vinegar for preparing pickles etc.
(c) It is used for preparing various organic compounds like acetone, acetic anhydride ester etc.
(d) It is used as cogulating agent in rubber industries.
(e) It is used for making perfumes and medicines.
UNIT TEST PAPER 8 — ORGANIC CHEMISTRY
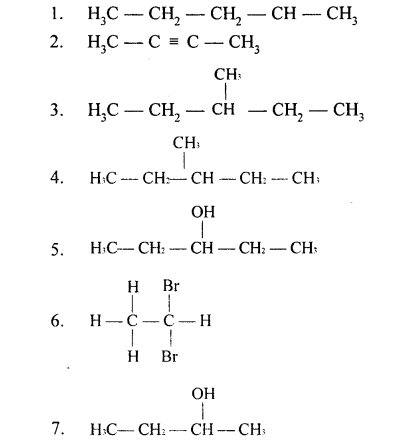
Question 1.
Draw the branched structural formula of the following organic compounds whose IUPAC names are given below.
- Pent-l-ene
- But-2-yne
- 3-methyl pentane
- 2-methyl-prop-l-ene
- Pentane-3-ol
- 1, 1, 2, 2 tetrabromoethane
- 2-methyl butan -2-ol
- 2, 2 dimethylpropan-l-ol
- 2, 2 dimethyl propane
- 2-bromo-4-chloro pentane
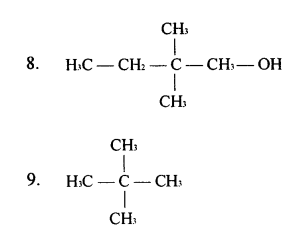
Ans.
Question 2.
Select the correct answer from the choice in brackets.
1. The vapour density of the fifth member of the homologous series of alkanes. [22 / 36 / 29]
2. The isomer of pentane which has ‘1’ C atom attached to ‘4’ other C atoms [n – / iso- / neo-] pentane.
3. The IUPAC name of the product of reaction of ethylene with hydrogen bromide, [ethyl bromide / bromoethane / dibromoethane]
4. The IUPAC name of methyl acetylene. [1-butyne / propyne / ethyne]
5. The functional group in ethanoic acid, [aldehydic / carboxyl / hydroxyl]
Ans:
- 36
- neo-pentane
- bromoethane
- propyne
- carboxyl
Question 3.
Give balanced equations for the following conversions.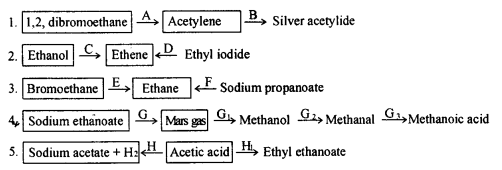
Answer:
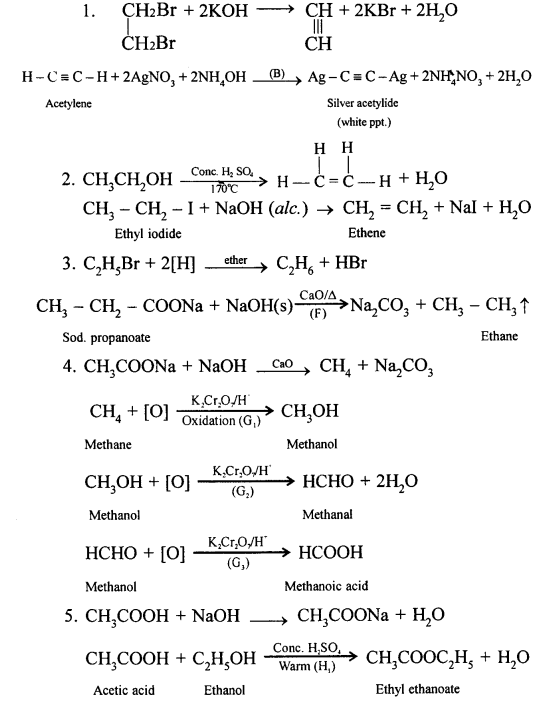
Question 4.
Select from the letters A to G the correct answer corresponding to the statements from 1 to 5 :
A :Ammoniacal CuCl2,
B : Trichloromethane,
C : Trichloroethane,
D : Bromine soln.,
E : Aqueous KOH
F : Ethene,
G : Sodalime,
H : Ethanol,
I : Ethyne.
- The organic compound which forms carbon tetrachloride on reaction with chlorine.
- The reagent which can distinguish between ethene and ethyne.
- The substance which reacts with bromoethane to give ethanol.
- The substance which gives bromoethane on reaction with hydrogen bromide.
- The substance which reacts with acetic acid to give CH3COOC2H5
Answer:
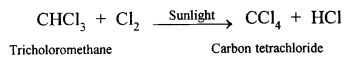
(1) -B
Explination
(2) – A
Explanation: [Only ethyne gives red ppt. of dicopper acetylide with ammoniacal Cu2Cl2 or CuCl]
(3) -E
Explination
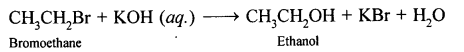
(4) -F
Eplination
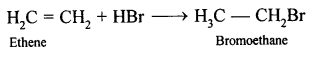
(5) -H
Explanation
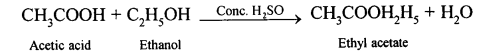
Question 5.
Give balanced equations for the following conversions.
- An alkyne to an alkene.
- An alkene to an alkane.
- An alkane to an alcohol.
- An alcohol to an alkene.
- A carboxylic acid to an ammonium salt
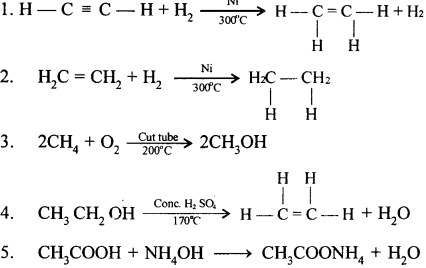
Question 6.
Give reasons for the following :
Question 6(1).
Concentrated sulphuric acid maybe added during esterification of acetic acid.
Answer:
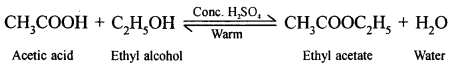
Cone. H2SO4, a strong dehydrating agent helps in the removal of water thus shifting the equilibrium in the forward direction, resulting the formation of more of ethyl acetate (ester).
Question 6(2).
Isomers belonging to the same homologous series may differ in physical properties but not in chemical properties.
Answer:
Isomers of the same homologous series have the same functional group (if any) and as such have similar chemical properties. As isomeres are different compounds they differ in one or more of their physical properties.
Question 6(3).
A given organic compound can be assigned only one name on the basis of the IUPAC system.
Answer:
This statement is not correct.Correct statement is : An organic compound may have more than one IUPAC name (out of all these one is a preferred IUPAC name) but two compounds cannot have the same IUPAC name because thismay lead to confusion.
Question 6(4).
Substitution reactions are characteristic reactions of saturated organic compounds only.
Answer:
Addition reactions are not possible in case of saturated organic compounds. Saturated organic compounds can only undergo substitution reactions.
Question 6(5).
Acetic acid is considered an aliphatic monocarboxylic acid.
Answer:
Acetic acid or ethanoic acid, CH3COOH has one carboxylic acid group (—COOH). Hence it is a monocarboxylic acid. As it has no benzene ring in it, it is not aromatic and hence it is an aliphatic monocarboxylic acid.
For More Resources
Hope given New Simplified Chemistry Class 10 ICSE Solutions – Organic Chemistry are helpful to complete your math homework.
If you have any doubts, please comment below. aplustopper try to provide online math tutoring for you.