How the Iron is made
IRON
Symbol | Fe | Atomic number | 26 |
Electronic Configuration of Iron :
The atomic number of iron is 26. This means that an atom of iron contains 26 electrons in its shells. The electronic configuration of iron is shown below.
| K | L | M | N | |
Fe(26) | 2 | 8 | 14 | 2 |
Thus, an atom of iron contains two electrons in its outermost shell.
Occurrence of Iron
Iron is second to aluminium in terms of abundance in the earth’s crust. It makes up 4.7% of the earth’s crust. Free iron has been found in most meteorites.
Iron is a reactive metal. So it does not occur free in nature. In combined state, it occurs as oxide, sulphide, carbonate, etc. The important ores of iron are:
(i) Haematite, Fe2O3
(ii) Magnetite, Fe3O4
(iii) Limonite, 2Fe2O3 . 3H2O
(iv) Siderite, FeCO3
(v) Iron pyrites, FeS2
The most important ore of iron is haematite, which is used most commonly in the extraction of iron. The pyrite ore (FeS2) is not used for the extraction of iron because of its high sulphur content.
Iron in India :
Iron metal has great economic importance. The world output of iron exceeds two hundred million tonnes per annum. In 2002-03, India’s total production of iron reached almost 97 million tonnes. Besides, India has a vast deposit of iron ore: about 12,318 million tonnes of haematite and 5,396 million tonnes of magnetite. Most of these deposits are located in Jharkhand, Orissa, Chhattisgarh, Tamil Nadu, Karnataka and Maharashtra. The important iron and steel plants are located at Bhillai, Bokaro, Jamshedpur, Rourkela, Durgapur, Asansol and Bhadravati.
Extraction of iron from Haematite :
1. Dressing of the ore :
The big lumps of the ore are broken into small pieces and then washed with water to remove clay, sand and other adhering impurities. The ore thus becomes ready for treatment in the blast furnace.
2. Smelting in the blast furnace :
The concentrated ore is mixed with coke and limestone. The mixture is charged at the top of a blast furnace. The following reactions occur in the furnace.
(i) As the charge comes down to the 873 K region, the iron oxide is reduced by the ascending carbon monoxide gas produced by the burning of coke.
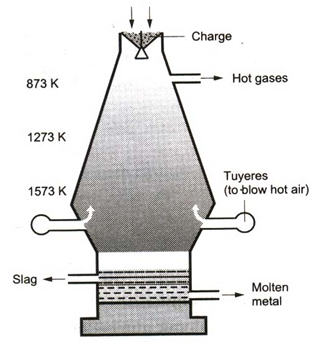
2C + O2 → 2CO
Fe2O3 + 3CO → 2Fe + 3CO2
The iron thus obtained is called sponge iron.
(ii) At the 1273 K region, the silica is converted to slag.
CaCO3 → CaO + CO2
CaO + SiO2 → CaSiO3
(iii) At the 1573 K region, sponge iron melts and dissolves carbon, phosphorus, silica, etc. The slag also fuses. The molten mass collects at the base of the furnace. The slag floats over it. The molten iron is taken out as required. This iron is called pig iron.
Function of limestone :Limestone is decomposed to give quicklime.
CaCO3 → CaO + CO2
Quicklime combines with impurities like sand to form a molten slag (calcium silicate).
CaO + SiO2 → CaSiO3
The slag floats on the surface of molten iron. It is taken out through a hole from time to time.
The formation of calcium silicate as slag not only removes unwanted silica but also keeps iron away from being oxidized.
Varieties of Iron :
1. Pig iron and cast iron : The iron produced in the blast furnace is pig iron. It contains a comparatively high percentage of carbon due to which it is hard and brittle. It also contains phosphorus, silicon and manganese as impurities. Pig iron is melted, mixed with steel scrap and allowed to cool in moulds to give cast iron. Cast iron is impure iron, and is hard and brittle.
2. Wrought iron : It is almost a pure form of iron. It contains only 0.12% to 0.25% carbon. It melts at a higher temperature (1773 K) than that at which cast iron melts. Wrought iron is obtained by melting cast iron on a hearth lined with ferric oxide (Fe2O3). The impurities such as carbon, phosphorus, silicon and manganese are oxidized by Fe2O3.
Fe2O3 + 3C → 2Fe + 3CO
Wrought iron is soft, grey and tough. It is malleable and ductile. Hence, it can be drawn into sheets and stretched into wires. It is used in making chains, wire, anchors and cores of electromagnets.
3. Steel : It is an alloy of iron and carbon. It contains about 0.15 to 1.7% of carbon. There are different types of steel.
(a) Mild steel : It contains less than 0.3% carbon. It is also called soft iron. Mild steel is used for making sheets and wires.
(b) Hard steel : It contains higher percentage (0.7-1.7%) of carbon. It is used in making tools and instruments.
(c) Alloy steels : Alloy steels are prepared by adding small amounts of nickel, cobalt, chromium, tungsten, molybdenum, manganese and silicon to steel. Alloy steels are used extensively in making rock-crushing machinery, helmets, armour plate, cutlery, springs, etc.
(d) Medium steel : It contains 0.3-D.7% carbon. It is hard and is used in making rails, bridges, etc.
Tempering :
The hardness and elasticity of steel can be controlled by heat treatment. The steel is heated to a temperature below redness. It is then cooled slowly. The process is called tempering of steel. It is used to bring the steel to a suitable state of hardness and elasticity.
Annealing of steel :
Hard steel can be softened by heating it to a high temperature and then allowing it to cool down slowly. This process is called annealing.
Quenching of steel :
Hard steel is heated to a high temperature. It is then suddenly cooled by plunging into oil or water. Steel becomes as hard and brittle as glass. Steel produced in this way is known as quenched steel and the process of making such steel is known as quenching or hardening of steel.
Properties of Iron
Physical properties :
Pure iron has a grey colour. It is malleable and ductile. It is a good conductor of heat and electricity. It melts at 1808 K and boils at 3023 K. It has a density of 7.9 × 103 kg cm–3.
Chemical properties :
1. Valency : Iron shows variable valency: 2 and 3. It forms divalent ion (Fe2+) as well as trivalent ion (Fe3+). The compounds in which iron shows divalency are known as ferrous compounds, whereas the compounds in which iron shows trivalency are known as ferric compounds. For example, in FeCl2 the valency of iron is 2. So, it is called ferrous chloride. In FeCl3, the valency of iron is 3. Hence, it is called ferric chloride.
2. Action of air : In the presence of moist air and carbon dioxide, iron gets covered with a thin deposit of rust. The rust consists of hydrated ferric oxide (2Fe2O3.3H2O).
3. Action of water : Red hot iron decomposes steam, forming ferrosoferric oxide and evolving hydrogen gas.
![]()
4. Action of acids : Iron lies above hydrogen in the activity series of metals. So, it can displace hydrogen from dilute hydrochloric acid or dilute sulphuric acid. The corresponding ferrous salts are also produced.
(i) With hydrochloric acid
(a) Iron dissolves in dilute hydrochloric acid, forming ferrous chloride with the evolution of hydrogen gas.
Fe + 2HCl → FeCl2 + H2
(b) Concentrated hydrochloric acid also produces hydrogen with iron.
(ii) With sulphuric acid
(a) Iron dissolves in dilute sulphuric acid, forming ferrous sulphate. Hydrogen gas is evolved in the reaction.
Fe + H2SO4 → FeSO4 + H2
(b) Iron reacts with concentrated sulphuric acid to form ferrous sulphate with the evolution of sulphur dioxide. Hydrogen gas does not evolve in this reaction.
Fe + 2H2SO4 → FeSO4 + SO2 + 2H2O
Some ferric sulphate is also formed due to the oxidation of FeSO4 by concentrated H2SO4,
2FeSO4 + 2H2SO4 → Fe2(SO4) + 2H2O + SO2
(iii) With nitric acid :
(a) Iron reacts with dilute nitric acid to form ferrous nitrate and ammonium nitrate.
4Fe + 10HNO3 → 4Fe(NO3)2 + NH4NO3 + 3H2O
(b) With concentrated nitric acid, iron is rendered passive due to the formation of insoluble ferrosoferric oxide (Fe3O4) on the surface of iron.
5. Action of halogens : Halogens combine with heated iron, forming the halides of iron. For example, chlorine combines with heated iron to form ferric chloride.
2Fe + 3Cl2 → 2FeCl3
6. Action with sulphur : When iron filings are heated with sulphur, iron sulphide is produced.
Fe + S → FeS
7. Displacement of less electropositive metal : When an iron piece is dipped into a solution of copper sulphate, copper is displaced from the salt and gets deposited on the surface of iron. This is because copper is less electropositive than iron.
Fe + CuSO4 → Cu + FeSO4
Tests to distinguish between ferrous and ferric salts
(i) When a ferrous salt solution is treated with a solution of sodium hydroxide, a greenish precipitate of ferrous hydroxide is obtained.
FeSO4 + 2NaOH → Fe(OH) + Na2SO4
When a ferric salt solution is treated with a solution of sodium hydroxide, a brown precipitate of ferric hydroxide is produced.
Fe2(SO4)3 + 6NaOH → 2Fe(OH) + 3Na2SO4
(ii) The ferrous salts are generally green-coloured, whereas the ferric salts are brown in colour.
Rusting :
When iron is exposed to moist air, a reddish-brown coating of a mixture of ferric oxide (Fe2O3) and ferric hydroxide ((Fe(OH3)) is deposited on the surface of the metal. This reddish-brown coating is known as rust, and this process is known as rusting. Thus, the slow conversion of iron into a mixture of Fe2O3 and Fe(OH) by water and atmospheric oxygen is known as rusting.
Rusting of iron is an oxidation reaction that occurs due to the attack of water and oxygen. It has been found that rusting does not take place in air-free water. It also does not occur in presence of oxygen alone. Both water and oxygen are essential for rusting. Thus, the following conditions are necessary for rusting:
(i) Presence of oxygen or air
(ii) Presence of water or moisture
The process of rusting is continuous. The strength of iron decreases gradually and finally the metal is destroyed completely.
Prevention of Rusting :
Iron can be prevented from rusting by keeping it out of contact with air and water, and also by converting it into an alloy. This can be achieved in the following ways.
- By covering the surface of iron with grease, paint, varnish, enamel, etc.
- By galvanizing iron: a thin coating of zinc is deposited on the surface of the iron object. This is done by electroplating. Since zinc does not corrode on exposure to air, zinc metal prevents iron from rusting.
- By coating the surface of the iron object with chromium, tin, nickel, or aluminium. These metals resist corrosion. Hence, they protect iron from rusting.
- By converting it into an alloy with chromium and nickel. This alloy is called stainless steel.
Uses of Iron :
- Iron is used in making household utensils and equipments.
- Wrought iron and cast iron are largely used in the manufacture of locomotives, railway lines, springs, tubes, etc.
- Iron finds wide application in house construction, e.g., in the reinforcement of roofs and other parts of buildings.