New Simplified Chemistry Class 8 ICSE Solutions – Language of Chemistry
ICSE SolutionsSelina ICSE SolutionsML Aggarwal Solutions
Simplified ChemistryChemistryPhysicsBiologyMathsGeographyHistory & Civics
EXERCISE
Question 1.
Explain the term ‘symbol’. State a reason why – the symbol of calcium is ‘Ca’ & of copper is ‘Cu’
Answer:
Symbol is a short form or abreviated name – of the element.
OR
“Symbol of an element is the ‘first letter’ or the ‘first letter and another letter’ of the English name or Latin name of the element”.
As the name calcium and copper start with same letter of English alphabet ‘C’ so another letter from the name of the element is added to ‘C’.
Hence symbol of calcium is ‘Ca’ and symbol of copper or cuprum (Latin name of copper) is ‘Cu’.
Question 2.
Define the term ‘valency’. With reference to water & ammonia as compounds respectively, state the valency of oxygen & nitrogen. Magnesium [2, 8, 2] has valency 2+. Give reasons.
Answer:
Valency is the number of hydrogen atoms which combine with (or displace) one atom of the element forming a Compound. Compound water H20, two atoms of hydrogen combine with one atom of oxygen to form water. Hence valency of oxygen atom is 2.
Compound ammonia NH3.
Valency of atom nitrogen is 3, as it combines with 3 atoms of hydrogen.
Valency of magnesium is 2+, Mg [2, 8, 2]
Electronic configuration of Mg is 2, 8, 2 i.e. there are 2 valence electrons which Mg can lose and achieve stable configuration of nearest noble gas Neon.
Hence is cation Mg2+ has valency 2+.
Question 3.
Explain the term ‘variable valency’. Copper having electronic configuration 2,8,18,1 exhibits variable valency. Give a reason for the same & name the compound CuCI & CuCl2.
Answer:
Variable valency : “When an element has more than one valency, its valency is called variable valency.”
Copper exhibit valency ‘1’ and ‘2’ i.e. has variable valency reason for variable valency :
Valency is – the number of electrons lost or gained from the outer shell of an atom of an element – during chemical reaction. Variation in this gain or loss results in ‘variable valency’.
Lower valency ends with – ous
Higher valency ends with – ic
CuCI is named as cuprous chloride (lower valency)
OR
Cu [I] Cl i.e. Copper [I] chloride and CuCl2 – Cupric chloride (higher valency) i.e. Copper [II] chloride Cu[II]Cl2
Question 4.
State the valencies of the following metallic elements –
(a) Potassium
(b) Sodium
(c) Calcium
(d) Magnesium
(e) Zinc
(f) Aluminium
(g) Chromium [write each symbol with the valency]
Answer:
Metals have positive valency 1, 2 or 3 ,
Valency of
(a) Potassium is K+
(b) Sodium 1+
Na+
(c) Calcium 2+
Ca2+
(d) Magnesium 2+
Mg2+
(e) Zinc 2+
Zn2+
(f) Aluminium 3+
Al3+
(g) Chromium 3+
Cr3+
Question 5.
Certain metals exhibit variable valencies which include valencies: 1+, 2+, 3+ & 4+.
State the variable valency of the following metals –
(a) Copper
(b) Silver
(c) Mercury
(d) Iron
(e) tin
(f) Lead
[write each symbol with the variable valency]
Answer:


Question 6.
State which of the following ions or radicals given below of non-metallic elements exhibit-valency: 1–, 2– & 3–
(a) Chloride (b) Bromide (c) Iodide (d) Nitrate (e) Hydroxide (f) Bicarbonate (g) Bisulphite (h) Bisulphate (i) Aluminate (j) Permanganate (k) Oxide (l) Sulphide (m) Sulphite (n) Sulphate (o) Carbonate (p) Dichromate (q) Zincate (r) Plumbite (s) Phosphate (t) Nitride
[write each ion or radical with the correct valency]
Answer:


Question 7.
Differentiate between the terms – ‘Ion’ & ‘radical’ with suitable examples.
Answer:
Ion “is an atom or a group of atoms carrying a positive or negative charge due to loss or gain of electrons.” e.g. cation Na+ and Cl1- is anion.
Radical “is group of atoms of elements that behaves like a single unit & show valency.”
Positive radical [NH H41+ ] Ammonium
Negative radical [HCO31- ], [NO31-] Bicarbonate Nitrate
Question 8.
Write the chemical formula of the following compounds in a step-by-step manner – (a) Potassium chloride (b) Sodium bromide (c) Potassium nitrate (d) Calcium hydroxide (e) Calcium bicarbonate (f) Sodium bisulphate (g) Potassium sulphate (h) Zinc hydroxide (i) Potassium permanganate (j) Potassium dichromate (k) Aluminium hydroxide (l) Magnesium nitride (m) Sodium zincate (n) Copper [II] oxide (o) Copper [I] sulphide (p) Iron [III] chloride (q) Iron [II] hydroxide (r) Iron [III] sulphide (s) Iron [III] oxide.
Answer:






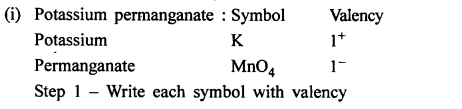

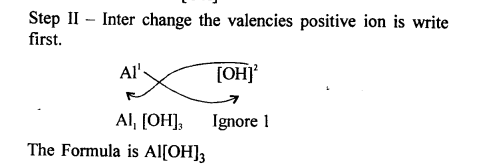

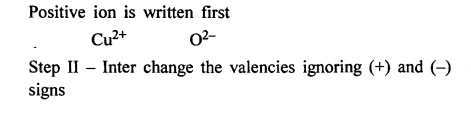

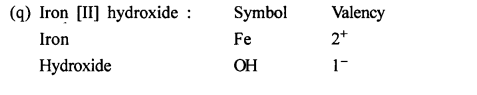

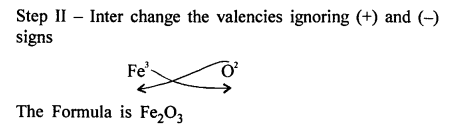
Question 9.
What’ is a chemical equation. How is it represented. Differentiate between a ‘word equation’ and a ‘molecular equation’ with a suitable example.
Answer:
“Chemical equation is a short form – representing the result of a chemical change.”
OR
“Is the representation of a chemical change.”
Word equation tells which substances react (take part) in chemical reaction and which substances are produced where as molecular equation symbols and molecular formulas are used for both reactants and products.
Example : When zinc reacts with dilute sulphuric acid, both being reactants, products produced are zinc sulphate and hydrogen gas, which are shown as below:
Word equation :
Zinc + Sulphuric acid → Zinc sulphate + Hydrogen
Molecular equation :
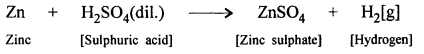
Question 10.
State the information provided by a chemical equation. Chemical equations suffer from a number of limitations. State the main limitations of a chemical equation.
Answer:
Information provided by a chemical equation :
(a) It tells us the formulas and symbols of the reactants and products.
(b) It tells us the ratio in which substances reacts or are produced. If limitations are covered.
(c) It tells the physical state of substances i.e. solid, liquid, gas.
(d) Whether the reaction is endothermic or exothermic.
(e) Conditions for starting the reaction i.e. if catalyst is needed or not.
(f) If reaction is reversible or not.
Limitations :
(a) Physical states of the reactants or products. But now we write along with substances (1) for liquids, (s) for solid and (g) for gas.
(b) Conditions that effect the reaction, i.e. temp, pressure or catalyst.
(c) Concentration of reactants and products we use dil. for dilute and cone, for concentrated.
(d) Nature of the chemical reaction.
(e) Speed – reaction is fast or slow.
(f) Exothermic or endothermic we write + heat or – heat towards products for exothermic and endothermic.
(g) The completion of the reaction.
Question 11.
State what is a balanced equation with a relevant example. Give a reason why an equation is balanced with reference to the law of conservation of matter.
Answer:
A balanced equation : “An equation is said to be balanced if the number of atoms of each element of the reactant is equal to the number of atoms of each element of the product.”
Example :
2Mg + O2 → 2MgO
[Reactants] [Product]
Reactans have 2 atoms of Mg and 2 atoms of oxygen.
Products have 2 atoms of magnesium and 2 atoms of oxygen.
∴ Number of atoms of each element of reactants = Number of atoms of each element of product.
- Reason for balancing equation : Law of conservation of matter says that “Matter is neither created nor destroyed during a chemical reaction.” This is possible only if number of atoms of each element on both sides of → are equal i.e. in reactants and also in products. Hence a reaction is balanced.
Question 12.
Write balanced molecular equations for the following word equations :


Answer:
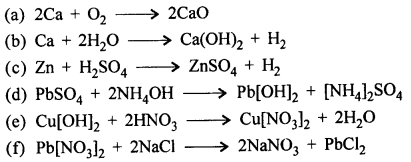
Question 13.
Balance the following equations :
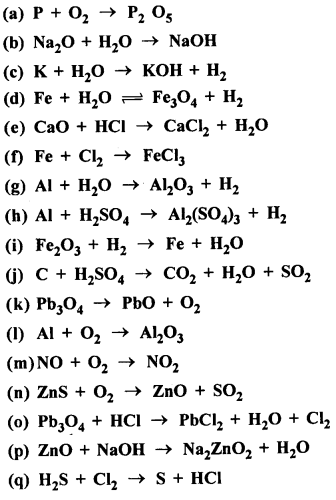
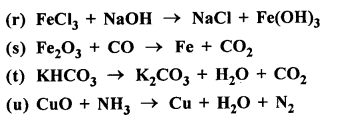
Answer:
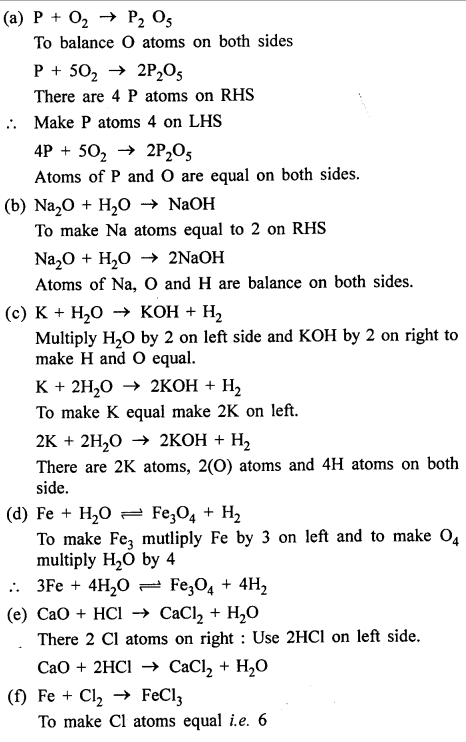
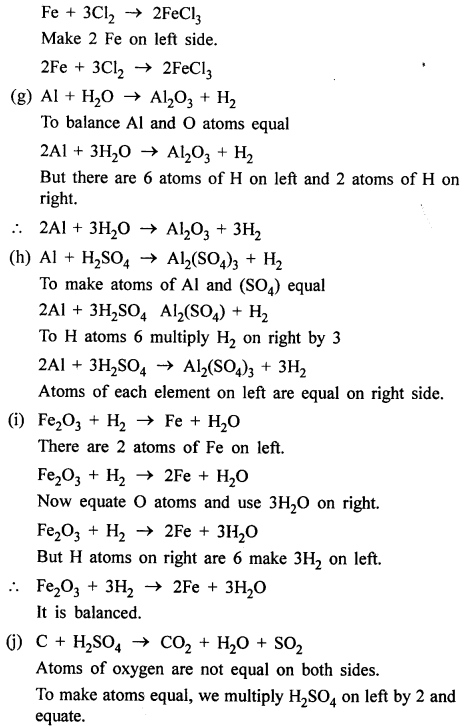
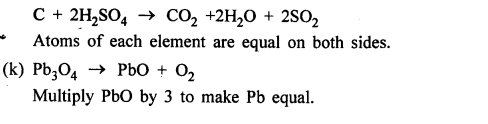

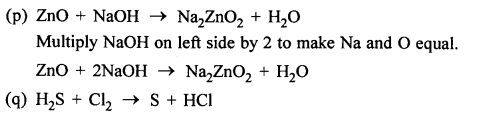

OBJECTIVE TYPE QUESTIONS
Q.1. Complete the statements given below by filling in the blank with the correct words.
Question 1.
The formula of silver [I] chloride is ___ [AgCl/AgCl2].
Answer:
The formula of silver [I] chloride is AgCl.
Question 2.
The basic unit of an element is a/an ___ [molecule/atom/ion]
Answer:
The basic unit of an element is a/an atom.
Question 3.
Atoms contains ___ [netron/nucleus/, with positively charged __ [electrons/protons].
Answer:
Atoms contains nucleus with positively charged protons.
Question 4.
Element ___ [calcium/lead/carbon] has the symbol derived from its Laltin name ‘plumbum’.
Answer:
Element lead has the symbol derived from its Laltin name ‘plumbum’.
Question 5.
From the elements – He, Br, Pt & O; the element which forms a polyatomic molecule is ___ & which is liquid at room temperature is ___
Answer:
From the elements – He, Br, Pt & O; the element which forms a polyatomic molecule is O & which is liquid at room temperature is Br.
Question 6.
The valency of iron in FeO is ___ [2+/1+] of chlorine [chloride] in CaCl2 is ___ [1–/2–] and of dichromate in K2 Cr2 O7 is [2+/2–].
Answer:
The valency of iron in FeO is 2+ of chlorine [chloride] in CaCl2 is 1– and of dichromate in K2 Cr2 O7 is 2–.
Q.2. Match the statements – 1 to 10 below with their correct answers from – A to J.

Answer:


Q.3. Match the compounds in List I – 1 to 20 with their correct formulas in List II – A to T.

Answer:

Q.4. Underline the incorrectly balanced compound in each equation & rewrite the correct equation.
Question 1.
2Na + 3H20 → 2NaOH + H2
Answer:
Correct equation is :
2Na + 2H20 → 2NaOH + H2
Question 2.
4P + 4O2 → 2P2O5
Answer:
Correct equation is :
4P + 5O2 → 2P2O5
Question 3.
FE2O3 + 2H2 → 2Fe + 3H20
Answer:
Correct equation is :
FE2O3 + 3H2 → 2Fe + 3H20
Question 4.
2A1 + 2H2SO4 → A12(SO4)3 + 3H2
Answer:
Correct equation is :
2A1 + 3H2SO4 → A12(SO4)3 + 3H2
Question 5.
![]()
Answer:
Correct equation is :
![]()
Question 6.
ZnO + 3NaOH → NA2ZnO2 + H20
Answer:
Correct equation is :
ZnO + 2NaOH → NA2ZnO2 + H20
Question 7.
FeCl3 + 3NH4OH → 2NH4C1 + Fe(OH)3
Answer:
Correct equation is :
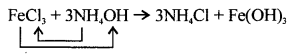
Question 8.
FeS + 2HCl → 2FeCl2 + H2S
Answer:
Correct equation is :
FeS + 2HCl → FeCl2 + H2S
Question 9.
3NH3 + H2So4 → (NH4)2So4
Answer:
Correct equation is :
2NH3 + H2SO4 → (NH4)2SO4
Question 10.
PbO2 + 4HCl → PbCl2 + H20 + Cl2
Answer:
Correct equation is :
PbO2+ 4HCl → PbCl2 + 2H20 + Cl2