New Simplified Chemistry Class 8 ICSE Solutions – Hydrogen
ICSE SolutionsSelina ICSE SolutionsML Aggarwal Solutions
Simplified ChemistryChemistryPhysicsBiologyMathsGeographyHistory & Civics
EXERCISE
Question 1.
State how hydrogen occurs in the free state. Name three compounds containing hydrogen in the combined state.
Answer:
- In free state : In traces in the earth’s crust and atmosphere and in volcanic gases. It is also found in huge amount in the interior of the sun.
- In combined state : In organic compounds, plant and animals, in acids, in water.
Question 2.
Starting from zinc how would you obtain hydrogen using
(a) Steam
(b) A dilute acid
(c) An alkali
[ Give balanced equations for each & name the product formed in each case other than hydrogen]. Name a metal which will not react with the reactants above to give hydrogen.
Answer:
To obtain hydrogen using :
(a) Steam : Metals above [H] in activity series of metals, i.e. Zn, Fe and Pb react with steam and form corresponding oxides.
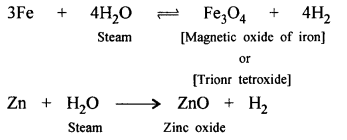
Metals below [H] do not react even with steam. Metal are Cu, Hg, Ag, Pt and Au.
(b) With dil. acids : Metals like Zn, Fe, Pb form corresponding salt.

Cu and metals below Cu do not react with dil. acids.
(c) An alkali : Zn, Pb react with concentrated alkali on boiling to give the corresponding metallic salt and liberate hydrogen.
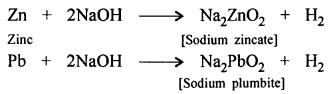
Question 3.
‘Hydrogen is obtained by electrolysis of acidified water’. Answer the following pertaining to the preparation of hydrogen by electrolysis, (a) The meaning of the term ‘electrolysis’ and ‘electrolyte’, (b) Name the electrode –
- through which the current enters the electrolyte.
- at which hydrogen is liberated.
Answer:
(a) Electrolysis : “Dissociation of water into hydrogen gas and oxygen gas by the passage of electric current is called electrolysis.”
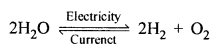
- Electrolyte : “A chemical compound (water) in solution state which conducts electric current and decomposes is called electrolyte.”
(b)
- Electrode through which current enters the electrolyte is anode.
- Electrode at which hydrogen is liberated is cathode.
Question 4.
In the laboratory preparation of hydrogen from zinc & dilute hydrocholoric acid – state a reason for
(a) Addition of traces of copper [II] sulphate to the reaction medium
(b) Collecting the hydrogen by downward displacement of water and not air & collecting it after all the air in the apparatus is allowed to escape
(c) Having the end of the thistle funnel dip below the level of the acid in the flask.
Answer:
(a) Addition of traces of copper [II] sulphate acts as catalyst and speeds up the reaction.
(b) Hydrogen is collected by downward displacement of water as it is slightly soluble in water and in air it form explosive mixture with air and also hydrogen is lighter than air.
(c) Otherwise the gas formed will escape through thistle funnel.
Question 5.
In the industrial method of preparation of hydrogen by the Bosch process – give
(a) Balanced equations for the first two main steps in the production of hydrogen
(b) The reason for use of addition of a promoter to the catalyst in the final step
(c) The name of the solution which absorbs the unreacted carbon monoxide.
Answer:
(a) Bosch process for industrial production of hydrogen :
- Step [I] : Preparation of water gas [CO + H2] by passing steam over white hot coke.
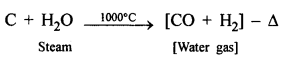
- Step [II] : Reduction of steam to hydrogen by carbon monoxide.
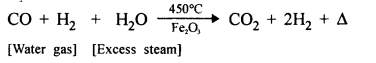
(b) Promotor Cr2O3 is used to increase the efficiency of catalyst [Fe2O3].
(c) Ammoniacal cuprous Chloride [CuCl] solution absorbs unreacted CO.
Question 6.
State the following pertaining to the physical properties of hydrogen :
(a) Colour & odour
(b) Solubility in water
(c) Effect on moist blue litmus paper.
Answer:
(a) Colour and odour of hydrogen gas colourless. Odourless.
(b) Slightly soluble in water.
(c) No-effect i.e. colour does not change.
Question 7.
Draw neat labelled diagrams for two different experiments to prove that – hydrogen is lighter than air.
Answer:
- To show that H2 is lighter than air :

- Air from jar B being heavier runs down in jar A and H2 runs to jar B as it is lighter than air and burns with ‘pop’ sound in jar B if a burning splinter is brought there.
Question 8.
Starting from hydrogen gas how would you obtain
(a) A neutral liquid
(b) A basic gas
(c) A metal by reduction of its heated oxide.
[The metal formed is above iron in the activity series]
Answer:
(a) Hydrogen bums quietly in oxygen to produce water (a neutral liquid)
2H2 + O2 → 2H20
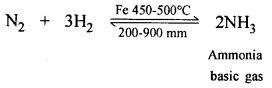
(b) 3 volumes of H2 react with 1 vol. of N2 gas in presence of catalyst (finely divided Fe at 450-500°C) and a basic gas ammonia is produced.
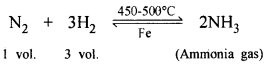
(c) Reduction of Fe2O3 (iron oxide) on heating to metal iron.

Question 9.
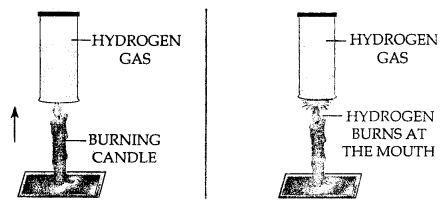
Using a burning candle and a jar of hydrogen – how would you prove experimentally that (a) Hydrogen is a combustible gas (b) Hydrogen does not support combustion.
Answer:
(a) Hydrogen gas is combustible and bums at the mouth of the gas jar. When a burning candle is brought near the mouth of hydrogen in gas jar.
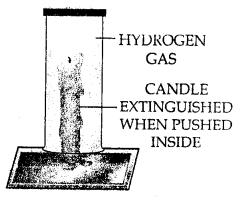
(b) Hydrogen extinguishes the burning candle when pushed inside the jar. This shows that hydrogen does not support combustion.
Question 10.
State a reason why, when hydrogen is passed over heated copper oxide, the resultant product formed, differs in colour from the original reactant.
Answer:
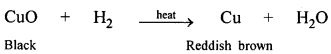
H2 removes oxygen from CuO and reduces it to Cu metal (reddish brown in colour).
Question 11.
With reference to the uses of hydrogen, give reasons for the following :
(a) Hydrogen is not used in air balloons
(b) A mixture of hydrogen & oxygen on burning, find application in welding & cutting metals
(c) Reaction of hydrogen with nitrogen under specific conditions finds industrial utility.
Answer:
(a) Hydrogen gas in ‘highly inflamable’ i.e. catches fire. Hence generally it is not used in air balloons.
(b) Hydrogen and oxygen mixture on burning produces a high temperature upto 2800°C which is used for welding and cutting of metals.
(c) One vol. of N2 and 3 vol. of H2 in presence of a catalyst (finely divided iron) at 450-500°C and at 200-900 mm pressure forms basic compound ammonia [NH3] which finds applications in fertilizers and nitric acid [HNO3] and explosives.
Question 12.
Give a test to differentiate between two gas jars – one containing pure hydrogen and the other hydrogen-air mixture.
Answer:
When a burning splinter is brought near the gas jar containing, pure H2 bums with pale blue flame quietly.
H2 + Oxygen mixture bums with explosion.
Question 13.
With reference to oxidation & reduction reaction – complete the statement given by filling in the blanks with only the words (a) Addition (b) Removal.
‘Oxidation is a chemical reaction involving ____ of oxygen to a substance or ____ of hydrogen from a substance. Reduction on the otherhand involves ____ of hydrogen to a substance or ____ of oxygen from a substance.
Answer:
‘Oxidation is a chemical reaction involving – addition of oxygen to a substance or removal of hydrogen from a substance. Reduction on the otherhand involves addition of hydrogen to a substance or removal of oxygen from a substance.
Question 14.
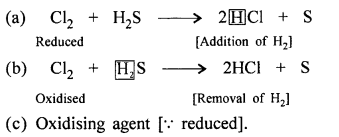
With reference to the equation : Cl2 + H2S → 2HCl + S pertaining to a redox reaction – select the correct answer in each case –
(a) Chlorine is oxidised/redcued to HCl.
(b) Hydrogen sulphide is oxidised/redcued to sulphur since the reaction involves addition/removal of hydrogen.
(c) Chlorine acts as an oxidising/reducing agent.
Answer:
OBJECTIVE TYPE QUESTIONS
Q.1. Give balanced equations for the following conversions :
Question 1.
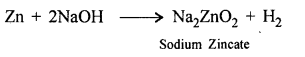
Zinc to sodium zincate – using an alkali.
Answer:
Zinc to sodium zincate.
Question 2.
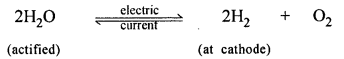
Acidified water to hydrogen – by electrolysis.
Answer:
Question 3.
Water gas to hydrogen – industrially.
Answer:
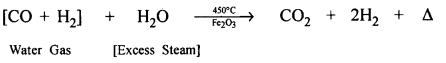
CO2 is removed by dissolving the mixture obtained in water under pressure
By passing the mixture through water or KOH [Caustic Potash Solution]
2KOH + CO2 → K2CO3 + H20
Residual gas left is HYDROGEN.
Question 4.
Iron [III] oxide to iron – using hydrogen.
Answer:
Fe2O3 is reduced by hydrogen

Question 5.
Nitrogen to a basic gas – using hydrogen.
Answer:
Nitrogen to basic gas [NH3] – using H2
Q.2. Give reasons for the following :
Question 1.
Copper does not displace hydrogen from dilute hydrochloric acid, but zinc does.
Answer:
Copper lies below [H] in activity series of metals and cannot displace hydrogen from acid.
Zn lies above [H] in reactivity series of metals and being more reactive can displace hydrogen from dil. acids.
Question 2.
In the preparation of hydrogen by electrolysis of water – the distilled water used is acidified.
Answer:
Pure or distilled water is bad conductor of electricity and does not form ions. To increase the number of H+ ions distilled water is acidified and made good conductor of electricity.
Question 3.
In the laboratory preparation of hydrogen from zinc and dilute hydrochloric acid – the zinc used granulated zinc.
Answer:
Granulated zinc contains traces of impurities which act as catalyst and increase the rate of production of hydrogen.
Question 4.
In Bosch process – the final gaseous products are passed through caustic potash [KOH] soln.
Answer:
In Bosch process, final products are passed through caustic potash solution to separate hydrogen from the mixture. CO2 dissolves in KOH leaving behind hydrogen.
Question 5.
The reaction of chlorine with hydrogen sulphide is deemed a redox reaction.
Answer:
Reaction of Cl2 with H2S is redox reaction as Cl2 gets reduced to HCl and H2S is oxidised simultaneously in the reaction. Hence it is redox reaction.
Q.3. Select the correct answer from A, B, C, D or E for each statement given below :
A: Nickel
B: Sodium
C: Iron
D: Iron [III] oxide
E: Magnesium oxide
Question 1.
A metal which reacts with water to give a metallic hydroxide & liberate hydrogen.
Answer:
B : Sodium
Question 2.
The metallic compound used as a catalyst in Bosch process.
Answer:
D : Iron [III] oxide
Question 3.
The metal used as a catalyst in hydrogenation of oils.
Answer:
A : Nickel
Question 4.
The metal which reacts with steam liberating hydrogen & the reaction is reversible.
Answer:
C : Iron
Question 5.
The metallic compound formed when a metal above aluminium in the activity series reacts with steam.
Answer:
E : Magnesium oxide
Q.4. Select the correct answer from the choice in bracket to complete each sentence.
Question 1.
The acid ___ [dil.H2SO4, dil. HNO3, dil. HCl] is not Used in the laboratory preparation of hydrogen, using zinc and an acid.
Answer:
The acid dil. HNO3 is not used in the laboratory preparation of hydrogen, using zinc and an acid.
Question 2.
In Bosch process the catalytic reduction of steam to hydrogen is carried out by ___ [CO2, CO, C].
Answer:
In Bosch process the catalytic reduction of steam to hydrogen is carried out by CO [carbon monoxide].
Question 3.
A foul smelling gas formed when hydrogen reacts with a molten non-metal, is ___ [hydrogen chloride, hydrogen sulphide, ammonia].
Answer:
A foul smelling gas formed when hydrogen reacts with a molten non-metal, is hydrogen sulphide [H2S].
Question 4.
The product formed on combustion of hydrogen in air is ___ [water gas, water, producer gas].
Answer:
The product formed on combustion of hydrogen in air is water.
Question 5.
The gas which has now replaced hydrogen in air balloons is ___ [argon, helium, neon].
Answer:
The gas which has now replaced hydrogen in air balloons is helium.
Q.5. Match the statements in List I with the appropriate answer in List II.

Answer:
