New Simplified Chemistry Class 7 ICSE Solutions – Metals and Non-metals
ICSE SolutionsSelina ICSE SolutionsML Aggarwal Solutions
Simplified ChemistryChemistryPhysicsBiologyMathsGeographyHistory & Civics
Points to Remember:
- Knowledge of chemistry plays a vital role in the development of human society and civilization.
- Metals are known to man from ancient times. Metals are used to make our life comfortable.
- Non-metals form another class of elements, hydrogen, oxygen, carbon, etc. They are used for various purposes.
- Alloys are homogeneous solid mixtures containing two or more metals e.g. steel, brass, bronze.
- Common salt, hydrochloric acid, carbohydrates, fats, proteins, vitamins, occur naturally and can also be prepared artificially.
- Fertilizers are artificially prepared substances, which are necessary for the proper growth of crops.
- There are a number of man-made materials that are used in our daily life for various purposes.- g., cement, plaster of pads, plastics.
- Medicines are used to cure diseases.
- Solution is a homogenous mixture of solute and solvent.
- Soda water is prepared by dissolving carbon dioxide in water under high pressure.
- Syrup is a highly concentrated sugar solutions. It contains a specific flavour.
Exercise
Question 1.
Give the common name and chemical name of an ore of –
(a) zinc (b) aluminium (c) iron.
Answer:

Question 2.
Distinguish between the physical properties of metals and non-metals with reference to –
(a) lusture (b) malleability (c) ductility (d) tensile strength (e) sonority (f) conduction of heat and electricity (g) melting and boiling point (h) density.
Answer:


Question 3.
With reference to the physical properties of metals and non-metals, state the following exceptions.
(a) a metal which is liquid at room temperature
(b) a non-metal which sublimes and is non-lustrous
(c) a non-metal which has low tensile strength
(f)) a non-metal which conducts electricity
(e) a metal which floats on water.
Answer:
(a) Mercury
(b) Iodine
(c) Phosphorus
(d) Graphite
(e) Lithium (Li), Sodium (Na), Potassium (k).
Question 4.
With reference to the following metals state their – use in daily life.Metals:
(a) copper (b) iron (c) aluminium (d) magnesium (e) zinc (f) lead (g) silver
Answer:
(a) Copper:
- Making calori meters as it is good conductor of heat.
- Making thin wires being conductor of electricity.
(b) Iron:
- Pig Iron is used in drain pipes as it is easily moulded into castings.
- Steel is used in Automobiles.
(c) Aluminium:
- Making utensils as a metal, it is easily moulded in utensils.
- In paints – Aluminium powder is mixed with linseed oil as it is anti-corrosive in nature.
(d) Magnesium:
- It is used in fire works as it bums with a dazzling light.
- Photography: Magnesium produces a bright flash. It is used in strip form.
(e) Zinc:
- For galvanising iron sheets because Zinc coated iron sheets does not rust.
- In Dry cells as Zinc container acts as cathode.
(f) Lead:
- Flexible pipes as it is malleable and reactant to corrosion.
- Bullets as it has high specific gravity and also is moulded into castings.
(e) Silver:
- For making Jewellery.
- As is conducts more electricity and heat than any other metal which makes it a prime source of use in electronics.
Question 5.
Describe a simple experiment to prove that – a copper wire conducts electricity, but a piece of coal does not.
Answer:
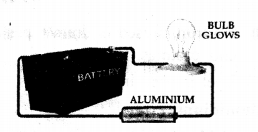
Experiment to prove that a copper wire conducts electricity
- Take a copper, battery, bulb and loose wire.
- Join the circuit i .e. wire with the battery, copper and bulb as shown in the figure.
- We will observe that the bulb glows, as metal i.e. copper is the good conductor of electricity.
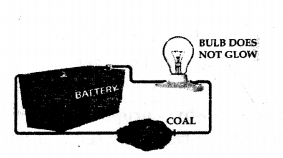
Experiment to prove that coal does not conducts electricity
- Take a coal, battery, bulb and wire.
- Join the circuit e. wire with battery, coal and bulb as shown in the figure.
- We will observe that the bulb will not glow, as non-metal — i.e. coal is bad conductor of electricity.
Question 6.
Differentiate between metals and non-metals with reference to –
(a) number of electrons in outer or valence shell (b) formation of cation and anion (c) reaction with dilute acids.
Answer:


Question 7.
Give a reason why – properties of metals are related to their activity or reactivity series of metals. Explain the same with reference to potassium and iron in the series.
Answer:
Activity of series of metals is a series of arrangement of metals in order of their reactivity.
Hence, the most active metal is at the top of the series & the least active metal is at the below of the series.
Therefore, the properties of metals are related to their activity or reactivity series of metals.
However, the Potassium (K) is at the top of the series. Hence, it reacts vigorously with cold water. The equation is
2K + H2O → 2KOH + H2
The Iron (Fe) is below the potassium, sodium, calcium and Magnesium. So, It reacts smoothly with water (steam). The equation is
![]()
Question 8.
Give the basic demarkation of the Modern Periodic Table into metals, metalloids and non-metals with special reference to halogens and noble gases.
Answer:
Modern Periodic Table is the arrangement of elements in increasing order of atomic numbers.
There are total 118 elements, the first element i.e. the atomic number 1 is Hydrogen and the last element i.e. the atomic number 118 is Ununoctium.
The elements are categorized mainly into 4 types, and these are:
- Reactive Metals: They are placed in the extreme left of the periodic table.
- Weak Metals: They are placed in the middle of the periodic table.
- Non-metals: They are placed in the upper right corner of the periodic table, and maybe subset further as halogens and noble gases.
- Metalloids: They have the properties of both the metals and non-metals.

The above demarkation may vary, since the nature of some elements is still under research.
Question 9.
Give a reason why the non-metals – halogens and noble gases are placed in separate groups of the periodic table with special reference to their difference in properties.
Answer:
Halogens and noble gases are placed in separate groups of the periodic table because halogens are extremely reactive elements because they need one more electron to gain a full octet of valence electrons, whereas the noble gases are extremely unstable because they already have their full octet.
Question 10.
State what are metalloids. Name the elements recognised as metalloids. Compare the properties of metalloids with those of metals. State the uses of three different metalloids.
Answer:
- Metalloids: Elements that show the properties of both metals and non-metals.They have physical and chemical properties, intermediate between those of metals and non-metals.
- Element recognized as metalloids: Commonly recognized – Boron [B], Silicon [Si], Germanium [Ge], Arsenic [As], Antimony [Sb] and Tellurium [Te] Least recognized metalloid is Polonium [PO],
- Comparing the Properties of Metalloids with those of Metals:
- State-
- Metals are solids at room temperature with the exception of mercury, which is liquid at room temperature.
- Metalloids are generally solids at room temperature
- Lustre-
- Metals have the quality of reflecting light from its surface and can be polished. Example Gold, Silver and Copper.
- They have metallic lustre and look like metals, but many show characteristic properties of weak non-metals.
- Conductivity –
- Metals are good conductors of heat and electricity.
- Metalloids are semi-conductors of electricity and show average transmission of heat.
- Malleability and ductile –
- Metals have the ability to withstand hammering and can be made into thin sheets known as foils.
- They are generally malleable and ductile. Silicon is not malleable or ductile, but and ductility is lustrous and acts as an electrical conductor under specific conditions.
Use of metalloids
They are too brittle, to have any structural uses in the pure form.
- Boron is used as an insecticide and a fire retardant.
- Silicon: Silicon gel is generally applied to bum patients and to absorb moisture.
- Arsenic: It is used for certain medicinal purposes.
- Antimony: It is used as an antiparasitic drug.
- Germanium: It finds application along with other metalloids in cell phones as a semiconductor.
Question 11.
Explain the term – rusting and give a word equation for the formation of rust. If polished iron nails are kept in three separate test tubes, state the contents in each test tube required, to prove the conditions for rusting.
Answer:
The term Rusting involves slow oxidation of iron in presence of air and moisture which results in the formation of rust i.e. hydrated iron [III] oxide.
THE OXIDATION REACTION
Rusting of Iron
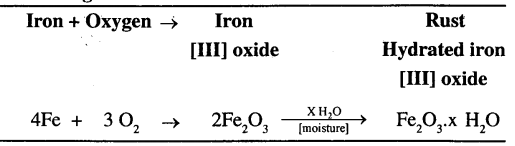
Iron when exposed to the atmosphere slowly reacts with the oxygen of the air, in the presence of moisture forming a brown coating called ‘rust’ on its surface. Rusting corrodes iron and reduces its structural strength.
Question 12.
State why telegraph poles are painted with aluminium paint.
Answer:
Aluminium paint is applied on telegraph poles because it act as an anti corrosive paint.
Question 13.
Give a reason why oiling or greasing is done on the exposed moving iron parts of machinery and not preferred on heavy stationary iron parts.
Answer:
Oiling or greasing is done on the exposed moving iron parts of machinery to avoid rusting and reducing friction whereas it is not preferred on heavy stationary iron parts as red lead oxide paint is used in heavy stationary iron.
Question 14.
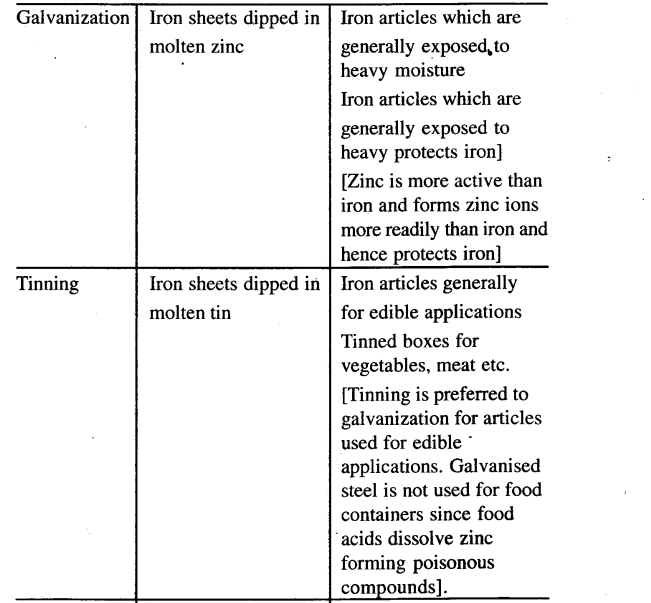
State why galvanization, tinning and chromeplating are used as three different processes for coating specific articles for prevention of rusting.
Answer:


Question 15.
Give a reason why –
(a) rust turns moist red litmus blue
(b) a green deposit is seen on the surface of an article of copper
(c) iron objects rust easily but the iron pillar at Qutab Minar has not rusted over the years.
Answer:
(a)
A suspension of rust is basic in nature and hence turn moist red litmus blue.
(b)
The green deposit of the surface of the articles made of copper is seen due to the formation of green copper carbonate.
(c)
The iron pillar at Qutab Minar has not rusted for 1600 years because of the following reasons:
- The high purity and great mass of iron may have created a temperature stabilizer thus reducing the condensation of moisture on it.
- The protective scale or passive iron coating formation on the surface, better forge welding and absence of impurities in iron – also may have dissuaded corrosion.
Question 16.
State a specific reason for using –
(a) Steel in automobiles
(b) gold in jewellery
(c) copper in electrical cables
(d) zinc in galvanizing
(e) lead in bullets
(f) magnesium in fireworks.
Answer:
(a) Steel is used in automobiles because it is malleable with high tensile strength.
(b) Gold is used in jewellery because it is expensive, lustrous, malleable and ductile.
(c) Copper is used in electrical cables because it is a good conductor of electricity.
(d) Zinc is used in galvanizing because it prevents rusting.
(e) Lead is used in bullets because it has a high specific gravity.
(f) Magnesium is used in making fireworks because it bums with a dazzling light.
Objective Type Questions
1. Complete the statements given below, by filling in the blank with the correct word/s:
- The ability of a metal to produce a reasonant sound sonority.
- A metal which floats on water potassium.
- A non-metal that leaves a mark on paper graphite
- A non-metal which allows an electric current to pass through it graphite
- A non-metal added to swimming pools to kill germs chlorine.
2. Give reasons for the following:
1. Metals form cations while non-metals form anions.
2. Potassium reacts vigorously with cold water while copper does not react with water or steam.
3. Rusting reduces the structural strength of iron.
4. A suspension of rust is basic in nature.
5. Galvanized steel is not used for articles used for edible applications.
Answer:
- Metals lose or donate valence electrons and form cations [positive ions], whereas Non-Metals gain or accept valence electrons and form anions [negative ions].
- This is because Potassium (K) is a most active metal and is at the top of the series whereas Copper (Cu) is a least active metal ans is at the below of the series.
- Rusting corrodes iron and hence reduces its structural strength.
- Rust is formed by oxidation of iron in air in presence of moisture. The main constitutent of rust is Iron(III) oxide [Fe2O3.xH2O] which is a metallic oxide. Hence, all metallic oxides are basic in nature. Therefore, a suspension of rust is basic in nature.
- Galvanised steel is not used for food containers because food acids dissolve zinc forming poisionous compounds.
3. Name the following:
- The most reactive halogen.
Ans. Fluorine. - A metal used in packaging.
Ans. Aluminium. - The chemical compound responsible for the green deposit on the surface of articles of copper.
Ans. Copper carbonate (CuCO3) - A metal used in flexible pipes.
Ans. Lead - A halogen which is a solid at room temperature.
Ans. Iodine
4. Match the correct answer from A, B, C, D and E – for each statement given below:
A : Boron B : Chlorine C : Potassium D: Platinum E: Radon
- The element placed in the extreme left of the Modern Periodic Table.
Ans. C : Potassium
- The element which shows properties of both metals and non-metals.
Ans. A: Boron.
- The element from the halogen family which is most abundant
Ans. B: Chlorine
- The element which is noble gas and radioactive in nature.
Ans. E: Radon
- The element which is a noble metal.
Ans. D: Platinum
5. State whether the statements given below are true or false. If false write the correct statement.
- Lead pencils contain lead.
False.
Correct: Lead pencils contain graphite.
- The chemical name of rust is iron [III] oxide.
True.
- Silicon is an example of a metalloid.
True
- Either oxygen or moisture is essential for rusting.
True - Graphite is a lustrous non-metal which conducts electricity.
True