New Simplified Chemistry Class 7 ICSE Solutions – Air and Atmosphere
ICSE SolutionsSelina ICSE SolutionsML Aggarwal Solutions
Simplified ChemistryChemistryPhysicsBiologyMathsGeographyHistory & Civics
Points to Remember:
- Air is a mixture of many gases, mainly Nitrogen = 78.1%, Oxygen = 20.9%, Carbon dioxide = 0.03 – 0.04%, Inert gases = 0.9%, [Water vapours, Dust particles and Impurities = Variable].
- Nitrogen is a colourless, an odourless and a tasteless gas. It is slightly lighter than air.
- The process of conversion of free atmospheric nitrogen into its compounds is called nitrogen-fixation.
- Oxygen constitutes about 21% of air by volume. It supports life on earth.
- Carbon dioxide is present in air in a very small quantity.i.e 0.03 – 0.04%. It is essential for the process of photosynthesis.
- Inert gases like neon, argon do not react with any substance.
- The harmful substances added to air are called pollutants.
- Some pollutants are suspended particles like pollen grains, oxides of sulphur and nitrogen, oxides of carbon, chlorofluorocarbons etc.
- Symbol of oxygen = O ; atomic number = 8, relative mass =16, molecule formula =O2
- Oxygen is available in free and combined state.
- A catalyst is a substance that increases or decreases the rate of a chemical reaction without itself undergoing any chemical change.
- Oxides are binary compounds formed by the chemical f combination of substance with oxygen.
- Rusting is the process in which iron slowly reacts with oxygen in the air and produces a flaky brown substance.
- Photosynthesis is a process by which CO2 and water are used up by green plants in the presence of sunlight to produce glucose and oxygen gases.
AIR & ATMOSPHERE
EXERCISE
1. State the meaning of the terms —
Question 1(a).
Air
Answer:
Air is matter which occupies space, has mass, exerts pressure and is highly compressible.
Question 1(b).
Atmosphere
Answer:
The atmosphere is a layer which extends about 300 kms above earth’s surface.
Question 2.
Name the important scientists and their studies which led to the discovery of the components of air.
Answer:
Discovery of air:
- John Mayow (1674) proved air has two components – active and inactive components.
- Lavoisier (1789) named active component as oxygen and inactive component as nitrogen
- Other scientists (1800’s) discovered noble gases, C02, water vapours.
Question 3.
Explain Lavoisier’s experiment which provided evidence to the discovery of components in air.
Answer:
Lavoisier’s experiment for the discovery of oxygen. Set up in apparatus as shown in the figure. When mercury oxide is heated in the retort, oxygen is collected in bell jar.
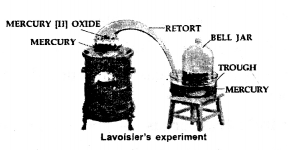
Observation:
- A red layer was formed in the heated mercury surface in the retort.
- The level of mercury in the trough rose by 1/5.
Conclusion: The active part of air removed by mercury on heating was named ‘oxygen’.The remaining inactive part of air in the bell-jar was named nitrogen.
Question 4.
Tabulate the various components of air, including the components with variable composition.
Answer:

Variation in composition of air:
- At high altitudes— The amount of oxygen is
Less – at high altitudes.
More – on the lower plains. - In deep seas — The amount of oxygen is-
Less – in deep seas and hence divers carry oxygen cylinders. - In industrial areas — The amount of impurities is-
More – in industrial and congested areas.
Question 5.
Compare the three main components of air e. nitrogen, oxygen and carbon dioxide with reference to their density and solubility, nature and reactivity.
Answer:

Question 6.
Of the total volume of air – 4/5th is nitrogen. State the utility or consequence of this ratio.
Answer:
The consequence of having 4/5 oxygen and 1/5 nitrogen in air would have been that earth would not have supported this kind of live and there would have been no balance of air and no photosynthesis.
Question 7.
State how nitrogen is utilised by plants for their growth. Give a reason why nitrogen is used in flushing food packages.
Answer:
Nitrogen of air is converted to soluble nitrogenous compounds in the soil in the presence of air and moisture. These-are absorbed by plants and converted to plant proteins.Nitrogen being inert or un reactive is filled into food packets to drive out the oxygen.
Hence bacterial growth inside the food package is reduced and thereby the food is well preserved and fresh.
Question 8.
Oxygen of the air is utilised for ‘combustiop’ and for ‘respiration’. Compare the two with a suitable examples.
Answer:
Oxygen is essential for burning of all components since it supports combustion, oxygen is also essential for respiration of all living things on the earth, it is utilised for respiration.
Burning of a candle involves oxidation of the carbon & hydrogen present in the candle resulting in the formation of carbon dioxide, water vapour and energy in form of heat and light.
Respiration involves oxidation of the carbon & hydrogen present in food materials resulting in formation of carbon dioxide, water vapour and energy in the form of heat.
Question 9.
State the utility of carbon dioxide for manufacture of food by plants. State its role in warming the earth’s environment and in excess resulting in global warming.
Answer:
Photosynthesis— It is a natural process by which green plants manufacture food in presence of sunlight.
Carbon dioxide in the atmosphere is absorbed by the plants in the presence of chlorophyll [the green pigment in plants] & sunlight, hence resulting in the formation of carbohydrates.
Green house effect— It consist of infrared, visible rays & ultra violet rays, which heat up the earth.
Carbon dioxide in the atmosphere allows the reflected ultraviolet rays to pass through, but prevents the infrared radiations from being radiated out of the earth’s surface. Hence, it results in warming of the earth’s environment.
Question 10.
State the utility of –
(a) Water vapour
(b) Rare or inert gases present in the air.
Answer:Utility of water vapour:
- For controlling evaporation.
- For determining climatic condition.
- For plant and animal growth.
The are called rare or noble gases because its composition is less than 1% in pure air.
Question 11.
Give the environment effect of three polluting gases with a reason why nitrogen is not considered a polluting gas.
Answer:
Environment effect of three different polluting gases
- Sulphur dioxide – causes respiratory problems
- Nitrogen dioxide – initiates acid rainfall
- Dust – causes respiratory problems
Oxygen and Nitrogen do not pollute environment. So, they are not polluting gases.
Question 12.
Describe simple experiments to show the presence of –
(a) oxygen and nitrogen component in air using a bell jar
(b) carbon dioxide in air using a test tube with outlets containing lime water.
(c) water vapour in air using a glass tumbler and ice.
Answer:
(a)
Oxygen and nitrogen in air:
Apparatus— A through, a bell jar, a cork and a crucible and a piece of white phosphorus.
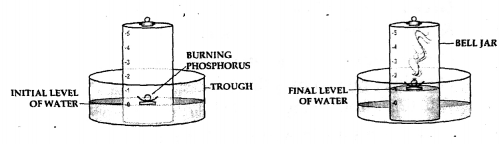
producer-
- The trough is filled with water and a bell jar divided into 5 equal parts, ‘1 to 5’ is placed over it.
- A crucible containing white phosphorus is placed on a cork which is made to float on water.
- The level of the water inside and out ride are adjusted to one level.
- The phosphorus is then ignited by means of a heated wire.
Observation—
- The phosphorus burns in the active component of air [oxygen] forming – dense white fumes of phosphorus pentoxide [P2O5]. Phosphorus + Oxygen →Phosphorus pentoxide
- The level of the water in the bell jar rises by l/5th.
Conclusion—
- Oxygen— The active component of air e. 1/5th pf air is used up in burning.
- Nitrogen— The inactive component of air e. 4/ 5 of air is not used up in burning. Hence air contains oxygen which supports combustion and nitrogen which does not.
(b)
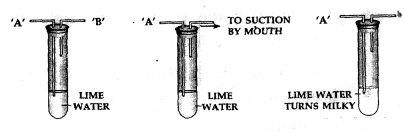
Carbon dioxide in air:
Apparatus— A test tube containing lime water with two outlets ‘A’ and ‘B’.
Procedure—Air is sucked slowly from the outlet ‘B’ as shown above.
Observation— The lime water in the test tube turns milky.
Conclusion— Air which is sucked in from end ‘A’, passes through the lime water and contains carbon dioxide which turns lime water milky [This is a test for carbon dioxide gas].
(c)
Water vapour in air:
Apparatus- A clean dry beaker ,cube of ice
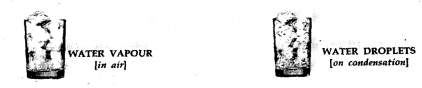
Procedure—
- Take a clean glass beaker dried from outside and place pieces of ice in it.
- Keep the beaker with the ice, aside for sometime.
Observation—
- Tiny droplets of water appear on the outer surface of the beaker, which can be proved by using a dry blue cobalt chloride paper. It turns pink in the presence of water.
Conclusion—
- The constituent of air-water vapour-
condenses on the cooler surface of the beaker, proving that air contains water vapour.
Question 13.
State four different properties of a compound and a mixture. With reference to the stated properties give reasons why air is considered a mixture and not a compound.
Answer:
Properties of compounds:
- Compound has a fixed composition.
- Compound has definite properties.
- Compounds cannot be separated by physical methods.
Properties of mixture:
- Mixture has a variable composition.
- Mixture has no definite properties.
- Mixtures can be separated by physical methods.
Air is a mixture because it has a variable, composition and has no definite properties.
Question 14.
Explain the term ‘air pollution’, with reference to air quality. State the sources of a few main pollutants.
Answer:
Air pollution:
Mixing of products (elements) from natural sources and man made sources to air, to make it unclean is called air pollution.
The various sources of air pollution are:
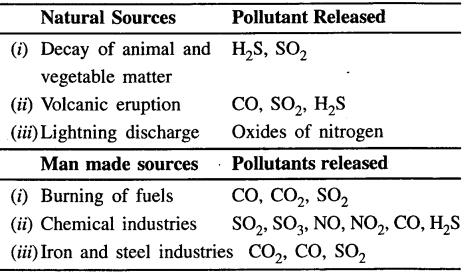
Question 15.
Identify the acidic oxides responsible for acid rain. State how their presence results in formation of acid rain. Give a reason why acid rain damages heritage buildings.
Answer:
Basic reactions for products formed in acid rain
- Sulphuric impurities in coal – form pollutant sulphur dioxide [SO2] and sulphur trioxide [SO3]
Sulphur dioxide + water vapour → Sulphurous acid [H2SO3]
Sulphur trioxide + water vapour→ Sulphuric acid [H2SO4] - Nitrogen in air at high temperatures form pollutant nitric oxide [NO] and nitrogen dioxide [NO2]
Nitric oxide + oxygen → nitrogen dioxide [NO2] Nitrogen dioxide + water → Nitrous acid [HNO2] and Nitric acid [HNO3]
Acid rain corrodes metallic surfaces & weakens building materials such as statues, marble, limestone [CaCO3] etc. It causes damage to heritage structures such as the Taj Mahal, which is predominantly made of lime stone and reacts with acids present in acid rain.
Question 16.
In the laboratory preparation of oxygen from hydrogen peroxide – answer the following:
(a) Draw a neat labelled diagram for the method used in the above preparation.
(b) Name the catalyst used in the preparation and state it’s function.
(c) State the word equation for the reaction involving the above preparation of oxygen.
(d) State the method of collection of the oxygen gas giving reasons.
Answer:
(a)
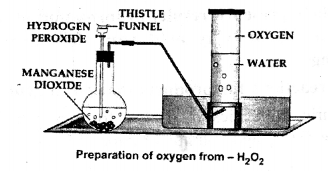
(b)
Catalyst used in preparation of oxygen is manganese dioxide. It increased the speed of decomposition of H2O2.
(c)

(d)
The method of collecting oxygen gas is by downward displacement of water.
The reasons are:
(1) Oxygen is very slightly soluble in water
(2) Oxygen is almost as heavy as air.
Question 17.
Give word equations for the reactions of –
(a) Non-metals – (1) sulphur (2) carbon (3) phosphorus and
(b) Metals – (1) sodium (2) calcium (3) magnesium with oxygen.
Answer:
(a) Non-Metals
(1)

(2)

(3)

(b) Metals
(1)

(2)

(3)

Question 18.
Distinguish between –
(a) Combustion and Respiration
(b) Combustion & Rusting with suitable word equations only ?
Answer:
(a) Combustion and Respiration
- Combustion involves oxidation of carbon & hydrogen in hydrocarbons.
- It is a rapid process at high temperature.
- The equation formed in Combustion is
Hydrocarbon + Oxygen → Carbon Dioxide + Water Vapour.
CxHy + O2 → CO2 + H2O - Where as Respiration is a process, whereby living things use oxygen from the air to oxidize food substances mainly glucose, in their body cells and release energy in form of heat.
- It is a slow process at body temperature.
- The equation formed in Respiration is
Glucose + Oxygen → Carbon Dioxide + Water Vapour + Heat.
C6H12O6 + 6O2 → 6CO2 + 6H2O + Heat energy
(b) Combustion and Rusting
Combustion
- It is confined to a variety of materials.
- Oxygen is required.
- Moisture is not essential.
- Oxide of carbon and hydrogen formed.
- Heat is evolved at a rapid rate and cause sudden rise in temperature.
Rusting
- It is confined only to iron.
- Oxygen is required
- Moisture is essential.
- Oxide of iron formed.
- Heat is evolved at a slow rate and cause slight rise in temperature
The equation formed in Combustion is
Hydrocarbon + Oxygen→ Carbon Dioxide + Water Vapour.
CxHy + O2 → CO2 + H2O
The equation formed in Rusting is Iron + Oxygen →Iron [III] oxide Rust Hydrated iron(III) oxide
![]()
Question 19.
Give the main experimental points only to demonstrate that – (a) magnesium (b) candle-gains weight on burning.
Answer:
(a) Experiment to show that magnesium gains weight on burning.
Take dry crucible with lid. Put a small piece of magnesium in it and cover the lid. Find its weight with lid covering it. Let it be 50 gms.
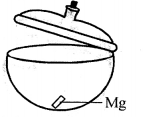
Now heat the crucible along with magnesium and lid just opened for a while. Taking care that no ash comes out. Let it cool covering the lid and products formed weight it again. The weight now will be more. Let it be 50.82 gms, oxygen combined is 50.82 – 50 = 0.82 gms this shows that something is added to magnesium, which has increased its weight. This 0.82 gm is the weight of oxygen added to magnesium.
2Mg + O2 → 2MgO
Hence magnesium gains weight on burning.
(b) Candle gains weight on burning
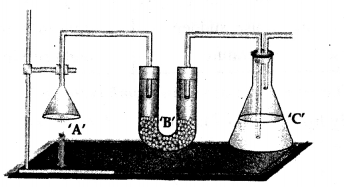
- Weight: The complete apparatus as shown above which includes –
- Unlit candle ‘A’.
- U tube ‘B’ containing anhydrous CaCl2.
- Conical flask ‘C’ containing cone. KOH soln.
Total wt. = a gms.
The candle ‘A’ is then lit and the products obtained on burning are allowed to be absorbed in U tube ‘B’ and conical flask ‘C’.
- Re-weigh: The complete apparatus as shown above after the candle has burnt – for a known period of time.
Total wt. = b gms.
Result:
- ‘b’ gms. is greater than ‘a’ gms.
- weight of apparatus after absorbtion
- of products is greater than the original weight of the apparatus
Conclusion: Candle gains weight due to mass of oxygen of the air which has combined with ‘carbon’ and ‘hydrogen’ of the candle [CxHy] producing .
- Water vapour [absorbed by anhydrous CaCl2]
- Carbon dioxide [absorbed by cone. KOH soln].
Question 20.
Give reasons for the following:
(a) patients suffering from lung problems are kept in an oxygen tent.
(b) oxygens finds utility in welding & cutting of metals.
Answer:
(a) Patients suffering from lung problems are kept in an oxygen tent for artificial supply of oxygen.
(b) Oxygen finds utility in welding and cutting of metals because of Oxy-acetylene flame provides temperature aroud 3000° C and oxy-hydrogen flame provides temperature around 2800°C.
Objective Type Questions
1. Match the statements in List I with the correct answer from List II.

Answer:

2. Give reasons for the following:
Question 1.
Acid rain has caused damage to heritage structures such as the Taj Mahal.
Answer:
Acid rain corrodes metallic surfaces & weakens the building materials such as statues, marble, lime stone [CaCO3] etc. It causes damage to heritage structures such as the Taj Mahal, which is predominantly made of lime stone and reacts with acids present in acid rain.
Question 2.
In the laboratory preparation of oxygen from potassium chlorate – manganese dioxide is added to the reactant.
Answer:
Because it alters the rate of the reaction – KC103 decomposes at a lower temperature.Catalyst itself undergoes no change –
(a) chemically (b) in mass.
Question 3.
Oxygen gas does not affect moist blue litmus paper, but carbon dioxide turns it slightly pink.
Answer:
Oxygen is neutral in nature, So, it does not affect blue litmus paper. Whereas Carbon dioxide is slightly acidic in nature, So, it changes blue litmus to slightly pink.
Question 4.
Oxidation of sulphur results in a product which turns moist blue litmus red.
Answer:
Oxidation of sulphur results in a formation of No’n-metallic acidic oxide i.e. Sulphur Dioxide (S02), hence moist blue litmus turns red.
Question 5.
Combustion and respiration show similarity.
Answer:
Oxidation is addition of oxygen or combination with oxygen.
Respiration involves oxidation of food (glucose) by inhaled oxygen to release C02, water vapour and heat energy. Burning also requires oxygen to produce C02 and heat energy.
3. Name the following:
- The product formed on oxidation of phosphorus.
Ans. Phosphorus pentaoxide. - The acid formed when the above product of oxidation of phosphorus, reacts with water.
Ans. Phosphorus acid. - The chemical name of rust.
Ans. Hydrated iron (III) oxide (Fe203-xH2O) - The pollutant which combines with water vapour to give sulphuric acid – a product of acid rain.
Ans. Sulphur trioxide. - The gas required for both combustion and rusting.
Ans. Oxygen
4. Select the correct answer from the choice in bracket to complete each sentence.
- Nitrogen of the air is utilized by plants for formation of proteins.
- The gas evolved as a product of combustion as well as respiration is carbondioxide.
- A candle burns in ajar of oxygen, but gets extinguished in nitrogen.
- Air has variable composition and hence there is less oxygen at lower altitudes than at higher altitudes.
- Rusting is a chemical reaction which involves slow oxidation.
5. Draw a neat labelled diagram for each of the following experiments.
Question 1.
To determine the component of air used up in burning, by using phosphorus and a bell jar.
Answer:
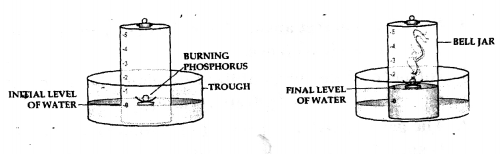
Question 2.
To study the presence of carbon dioxide in air.
Answer:
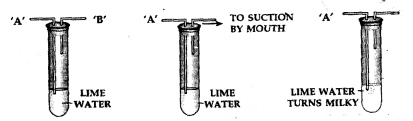
Question 3.
To prove that oxygen is non-combustible, but supports combustion.
Answer:
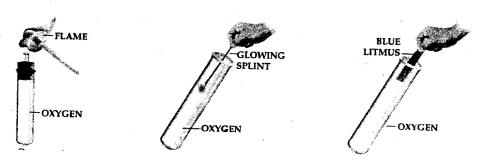
Question 4.
To prepare oxygen in the laboratory from hydrogen peroxide and a catalyst.
Answer:
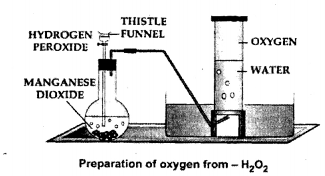
Question 5.
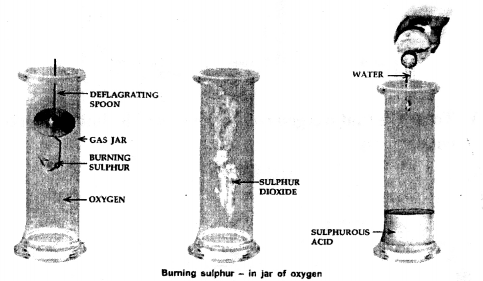
To demonstrate the burning of sulphur in a jar of oxygen.
Answer: